추천 제품
분석
98%
refractive index
n20/D 1.559 (lit.)
bp
250 °C (lit.)
mp
2-5 °C (lit.)
density
1.065 g/mL at 25 °C (lit.)
SMILES string
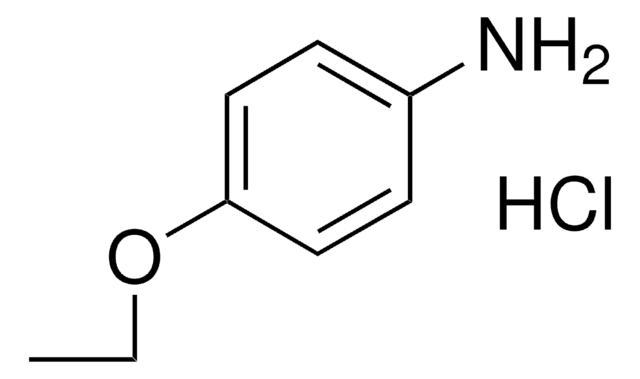
CCOc1ccc(N)cc1
InChI
1S/C8H11NO/c1-2-10-8-5-3-7(9)4-6-8/h3-6H,2,9H2,1H3
InChI key
IMPPGHMHELILKG-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Irrit. 2 - Muta. 2 - Skin Sens. 1
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 1
Flash Point (°F)
251.6 °F - closed cup
Flash Point (°C)
122 °C - closed cup
개인 보호 장비
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
T Lindqvist et al.
Chemical research in toxicology, 4(4), 489-496 (1991-07-01)
4-Ethoxyaniline (p-phenetidine) is oxidized by peroxidases to form several products, one of which is 4-[(4-ethoxyphenyl)imino]-2,5-cyclohexadien-1-one (1). This compound reacts with N-acetylcysteine (NAC) in methanol-phosphate buffers, generating at least four different products. Four major products, 4-[(4-ethoxyphenyl)amino]phenol (2), 3-(N-acetylcystein-S-yl)-4-[(4-ethoxyphenyl)amino]phenol (3), 2,5-bis(N-acetylcystein-S-yl)-4-[(4-ethoxyphenyl)-amino]phenol (4)
R Larsson et al.
The Journal of pharmacology and experimental therapeutics, 235(2), 475-480 (1985-11-01)
The metabolism of p-phenetidine in microsomes from rabbit kidney and the metabolism of acetaminophen and p-phenetidine in human kidney microsomes to protein binding metabolites were examined. Microsomal preparations from rabbit kidney medulla catalyzed the irreversible arachidonic acid-dependent binding of p-[14C]phenetidine
Annabel Kuek et al.
Postgraduate medical journal, 83(978), 251-260 (2007-04-04)
Targeted biologic therapies have revolutionised treatment of immune-mediated inflammatory diseases (IMIDs) due to their efficacy, speed of onset and tolerability. The discovery that clinically unrelated conditions, such as rheumatoid arthritis and Crohn's disease, share similar immune dysregulation has led to
R Larsson et al.
Chemico-biological interactions, 60(3), 317-330 (1986-12-01)
The interaction of N-(4-ethoxyphenyl)p-benzoquinone imine (NEPBQI), a metabolite formed during peroxidase catalyzed metabolism of p-phenetidine, with GSH and its effects in isolated rat hepatocytes were investigated. When reacted with GSH NEPBQI formed both a mono- and a diglutathione conjugate as
J D Baty et al.
Journal of chromatography, 353, 329-337 (1986-02-26)
Liquid chromatographic methods were developed for the study of the in vitro acetylation of the sulphonamide drug sulphamethazine and a series of aniline derivatives. The sensitivity of the methods have allowed data on the activity of the N-acetyltransferase enzyme(s) in
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.