추천 제품
Grade
certified reference material
pharmaceutical secondary standard
Quality Level
Agency
traceable to BP 368
traceable to Ph. Eur. G0326000
API family
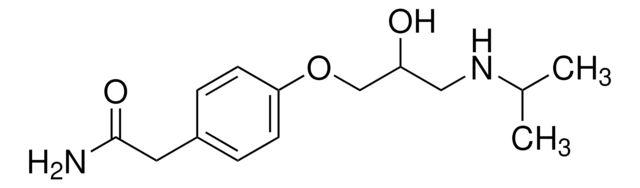
gliclazide
CofA
current certificate can be downloaded
기술
HPLC: suitable
gas chromatography (GC): suitable
응용 분야
pharmaceutical (small molecule)
형식
neat
저장 온도
2-8°C
InChI
1S/C15H21N3O3S/c1-11-5-7-14(8-6-11)22(20,21)17-15(19)16-18-9-12-3-2-4-13(12)10-18/h5-8,12-13H,2-4,9-10H2,1H3,(H2,16,17,19)
InChI key
BOVGTQGAOIONJV-UHFFFAOYSA-N
일반 설명
Gliclazide is an orally administered antidiabetic medication used in the treatment of type 2 diabetes mellitus, mainly for elderly patients.
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
애플리케이션
Gliclazide may be used as a pharmaceutical reference standard for the quantification of the analyte in pharmaceutical formulations using spectrophotometric technique and spectrofluorimetric technique.
These Secondary Standards are qualified as Certified Reference Materials. These are suitable for use in several analytical applications including but not limited to pharma release testing, pharma method development for qualitative and quantitative analyses, food and beverage quality control testing, and other calibration requirements.
분석 메모
These secondary standards offer multi-traceability to the USP, EP (PhEur) and BP primary standards, where they are available.
기타 정보
This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.
각주
To see an example of a Certificate of Analysis for this material enter LRAC3531 in the slot below. This is an example certificate only and may not be the lot that you receive.
관련 제품
제품 번호
설명
가격
신호어
Warning
유해 및 위험 성명서
예방조치 성명서
Hazard Classifications
Acute Tox. 4 Oral
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
이미 열람한 고객
Spectrophotometric determination of gliclazide in pharmaceuticals and biological fluids through ternary complex formation with eosin and palladium (II)
El-Enany N
Il Farmaco (Societa Chimica Italiana : 1989), 59(1), 63-69 (2004)
Spectrofluorimetric and spectrophotometric determination of gliclazide in pharmaceuticals by derivatization with 4-chloro-7-nitrobenzo-2-oxa-1, 3-diazole
El-Enany N
Journal of AOAC (Association of Official Analytical Chemists) International, 86(2), 209-214 (2003)
M V Shestakova
Vestnik Rossiiskoi akademii meditsinskikh nauk, (1)(1), 45-49 (2012-05-04)
Diabetic nephropathy (DN)--is a leading cause of terminal renal failure in the developed countries. Kidney pathology development undergoes several stages--from preclinical structural changes in the first years of the disease to diffuse or nodular glomerulosclerosis 15-20 years after the diabetes
Yige Bao et al.
European journal of pharmacology, 690(1-3), 142-148 (2012-07-07)
Recent studies have found that blockers of sulfonylureas receptor 1(SUR1) might have cardiac ischemic protective effects. We evaluated the effects of a selective SUR1 blocker gliclazide on cardiac function and arrhythmia after isoprenaline-induced myocardial injury in obese rats. Diet-induced obese
P C Lim et al.
The Medical journal of Malaysia, 67(1), 21-24 (2012-05-16)
Type-2 diabetes mellitus (T2DM) patients who were on gliclazide co-administered with metformin were changed to pre-combined glibenclamide-metformin tablets in the Endocrine Clinic, Penang Hospital. We conducted a retrospective study to evaluate the differences in glycaemic control and treatment cost following
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.