PHR1817
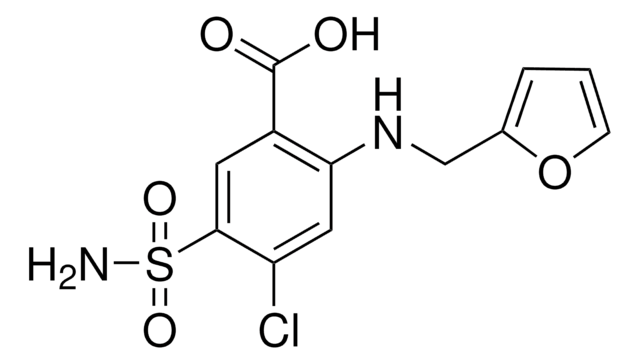
Furosemide Related Compound A
Pharmaceutical Secondary Standard; Certified Reference Material
동의어(들):
2-Chloro-4-furfurylamino-5-sulfamoylbenzoic acid, 2-CHLORO-4-N-FURFURYLAMINO -5-SULFAMOYLBENZOIC ACID
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
실험식(Hill 표기법):
C12H11ClN2O5S
CAS Number:
Molecular Weight:
330.74
Beilstein:
1297887
MDL number:
UNSPSC 코드:
41116107
NACRES:
NA.24
추천 제품
Grade
certified reference material
pharmaceutical secondary standard
Quality Level
Agency
traceable to Ph. Eur. F0702000
traceable to USP 1287020
CofA
current certificate can be downloaded
포장
pkg of 100 mg
mp
197 °C (dec.) (lit.)
응용 분야
pharmaceutical
형식
neat
저장 온도
2-30°C
SMILES string
OC(C1=C(Cl)C=C(NCC2=CC=CO2)C(S(N)(=O)=O)=C1)=O
InChI
1S/C12H11ClN2O5S/c13-9-5-10(15-6-7-2-1-3-20-7)11(21(14,18)19)4-8(9)12(16)17/h1-5,15H,6H2,(H,16,17)(H2,14,18,19)
InChI key
UXOOVYKVEXGCSH-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Pharmaceutical secondary standards for application in quality control provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards
애플리케이션
- Furosemide Impurity A for Quality Control: Furosemide Related Compound A serves as a crucial standard in pharmaceutical quality control, ensuring the purity and safety of furosemide as an active pharmaceutical ingredient. This impurity standard is essential for regulatory compliance and batch-to-batch consistency in drug manufacturing (Abasilim et al., 2023).
- Research on Drug Resistance: Furosemide Related Compound A is utilized in genomic research to identify potential drug resistance mechanisms. This research aids in understanding how bacterial pathogens develop resistance to drugs like clarithromycin and rifampicin, which is crucial for developing new therapeutic strategies (Mofidifar et al., 2024).
- Nanotechnology in Drug Delivery: High-purity Furosemide Related Compound A is used in the preparation and characterization of novel drug delivery systems, such as nanoparticles designed to enhance the bioavailability and efficacy of pharmaceutical compounds. This research highlights its application in improving drug formulations and targeting specific disease sites (Zeng et al., 2024).
- Impact on Bone Health: Research involving Furosemide Related Compound A explores its effects on bone health, particularly in how it influences vitamin D metabolism and bone density. This is vital for developing treatments that mitigate bone density loss associated with long-term use of diuretics like furosemide (Melanie KF et al., 2024).
- Nutritional Studies in Cancer Therapy: Furosemide Related Compound A is also examined for its role in nutritional interventions in cancer therapy, particularly its impact on the nutritional status of patients undergoing surgery for colorectal cancer. These studies are crucial for understanding how nutritional support can enhance recovery and treatment outcomes in cancer patients (Ural et al., 2024).
분석 메모
These secondary standards offer multi-traceability to the USP, EP and BP primary standards, where they are available.
기타 정보
This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.
각주
To see an example of a Certificate of Analysis for this material enter LRAC2891 in the Documents slot below. This is an example certificate only and may not be the lot that you receive.
추천 제품
Find a digital Reference Material for this product available on our online platform ChemisTwin® for NMR. You can use this digital equivalent on ChemisTwin® for your sample identity confirmation and compound quantification (with digital external standard). An NMR spectrum of this substance can be viewed and an online comparison against your sample can be performed with a few mouseclicks. Learn more here and start your free trial.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
이미 열람한 고객
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.