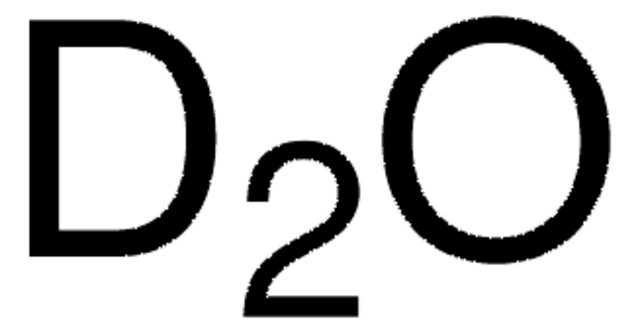151823
Chloroform-d
99.8 atom % D
Synonym(s):
Deuterochloroform
About This Item
Recommended Products
isotopic purity
99.8 atom % D
Quality Level
assay
≥99% (CP)
form
liquid
technique(s)
NMR: suitable
refractive index
n20/D 1.444 (lit.)
bp
60.9 °C (lit.)
mp
−64 °C (lit.)
density
1.500 g/mL at 25 °C (lit.)
mass shift
M+1
Looking for similar products? Visit Product Comparison Guide
General description
Application
Recommended products
recommended
suggested gloves for splash protection
signalword
Danger
Hazard Classifications
Acute Tox. 3 Inhalation - Acute Tox. 4 Oral - Carc. 2 - Eye Irrit. 2 - Repr. 2 - Skin Irrit. 2 - STOT RE 1 Oral - STOT SE 3
target_organs
Central nervous system, Liver,Kidney
Storage Class
6.1D - Non-combustible acute toxic Cat.3 / toxic hazardous materials or hazardous materials causing chronic effects
wgk_germany
WGK 3
flash_point_f
does not flash
flash_point_c
does not flash
ppe
Eyeshields, Faceshields, Gloves
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Use this reference table to find the coupling values and chemical shifts of our NMR (deuterated) solvents. Melting and boiling points, molecular weight, density, and CAS number are also listed.
Substances are said to be miscible in one another if they dissolve to form a uniform solution. Bookmark or download our miscibility table for common lab solvents.
Related Content
NMR spectroscopy elucidates molecular structure and purity via nuclear spin states in a strong magnetic field.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service