155756
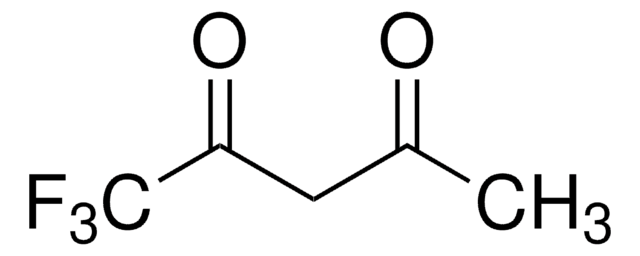
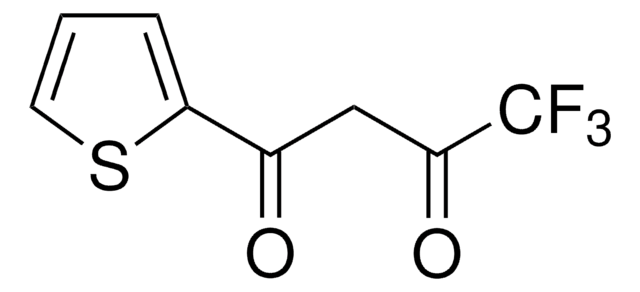
2,2,6,6-Tetramethyl-3,5-heptanedione
98%
Synonym(s):
Dipivaloylmethane
About This Item
Recommended Products
Quality Level
assay
98%
form
liquid
refractive index
n20/D 1.459 (lit.)
bp
72-73 °C/6 mmHg (lit.)
density
0.883 g/mL at 25 °C (lit.)
functional group
ketone
SMILES string
CC(C)(C)C(=O)CC(=O)C(C)(C)C
InChI
1S/C11H20O2/c1-10(2,3)8(12)7-9(13)11(4,5)6/h7H2,1-6H3
InChI key
YRAJNWYBUCUFBD-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
2,2,6,6-Tetramethyl-3,5-heptanedioneis a stable, anhydrous reagent. It undergoes O-additions and C-additions. In various reactions, it acts as an air-stable ligand for metal catalysts. Furthermore, it serves as a substrate for heterocycles.
Application
2,2,6,6-Tetramethyl-3,5-heptanedione used as a ancillary ligand in the synthesis of orange-emitting iridium(III) complex.
Storage Class
10 - Combustible liquids
wgk_germany
WGK 1
flash_point_f
152.6 °F - closed cup
flash_point_c
67 °C - closed cup
ppe
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Buchwald phosphine ligands for C-C, C-N, and C-O bond formation.
Buchwald phosphine ligands for C-C, C-N, and C-O bond formation.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service