All Photos(1)
About This Item
Empirical Formula (Hill Notation):
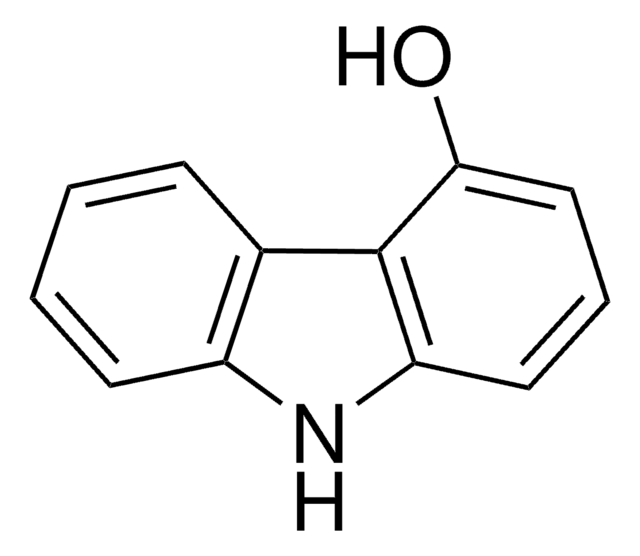
C12H9NO
CAS Number:
Molecular Weight:
183.21
Beilstein/REAXYS Number:
135859
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
97%
form
solid
mp
270-273 °C (lit.)
SMILES string
Oc1ccc2c(c1)[nH]c3ccccc23
InChI
1S/C12H9NO/c14-8-5-6-10-9-3-1-2-4-11(9)13-12(10)7-8/h1-7,13-14H
InChI key
GWPGDZPXOZATKL-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
2-Hydroxycarbazole is a compound structurally related to the Ca2+-mobilizing marine toxin, 9-methyl-7-bromoeudistomin. Room temperature electronic absorption and fluorescence spectra of 2-hydroxycarbazole has been studied in concentrated aqueous potassium hydroxide solutions. It undergoes chemoselective N-alkylation using NaH as a base in a THF-DMF solvent system.
Application
2-Hydroxycarbazole was used in the synthesis of isochromene fused carbazol, (4aS,13bR)-2,5,5-trimethyl-3,4,4a,5,8,13b-hexahydroisochromeno[3,4-b]carbazole.
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 2
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Nguyen Manh Cuong et al.
Natural product communications, 4(7), 921-924 (2009-09-08)
The first synthesis of isochromene fused carbazols, (4aS, 13bR)-2,5,5-trimethyl-3,4,4a,5,8,13b-hexahydroisochromeno[3,4-b]carbazole (2) and its epi-isomer 3 by condensation of citral and 2-hydroxycarbazole using Ti(OEt)4 and MeAlC12 as catalysts is described.
Tamanna Mallick et al.
Colloids and surfaces. B, Biointerfaces, 172, 440-450 (2018-09-10)
Six structurally different carbazoles (1-6) were explored as the green reducing agents for the synthesis of the fluorescent Au nanoparticles with tailor-made morphology in anionic (sodium dodecyl sulphate, SDS), cationic (cetyltrimethylammonium bromide, CTAB) and neutral (polyvinylpyrrolidone, PVP) micelle medium. Structure
K Zawadzka et al.
Environmental science and pollution research international, 22(24), 19658-19666 (2015-08-16)
Nitrogen heterocyclic compounds, especially carbazole, quinolone, and pyridine are common types of environmental pollutants. Carbazole has a toxic influence on living organisms, and the knowledge of its persistence and bioconversion in ecosystems is still not complete. There is an increasing
Chemoselective N-alkylation of 2-hydroxycarbazole as a model for the synthesis of N-substituted pyrrole derivatives containing acidic functions.
Albanese D, et al.
Tetrahedron, 51(19), 5681-5688 (1995)
J M Thomas et al.
The Journal of pharmacology and experimental therapeutics, 298(2), 644-650 (2001-07-17)
2-hydroxycarbazole, a compound structurally related to the Ca2+-mobilizing marine toxin 9-methyl-7-bromoeudistomin, has recently been proposed to activate both type 1 and type 2 ryanodine receptors in skeletal and cardiac muscle, respectively. This study was undertaken to evaluate the activity of
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service