221015
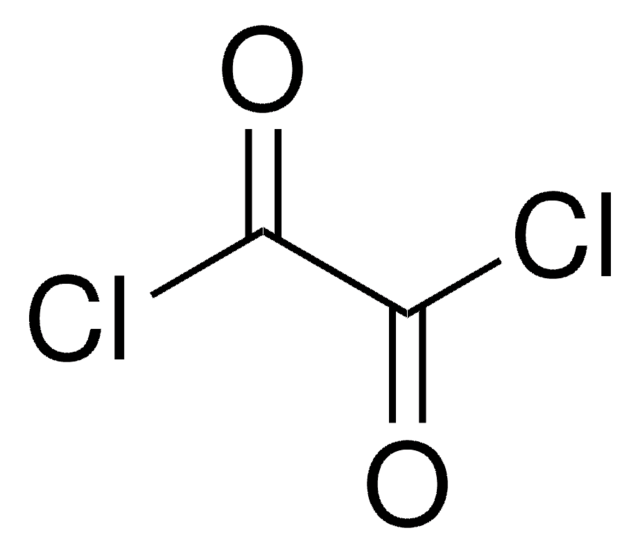
Oxalyl chloride
ReagentPlus®, ≥99%
Synonym(s):
Ethanedioyl dichloride
Select a Size
Select a Size
About This Item
Recommended Products
vapor density
4.4 (vs air)
Quality Level
vapor pressure
150 mmHg ( 20 °C)
product line
ReagentPlus®
assay
≥99%
form
liquid
reaction suitability
reagent type: oxidant
impurities
<10 ppb Heavy metals
color
APHA: 0-150
refractive index
n20/D 1.429 (lit.)
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- Preparation of Mosher′s acid chloride by reacting with Mosher′s acid in the presence of DMF.[2]
- Activation of dimethyl sulfoxide for use in the oxidation of long-chain alcohols to carbonyls.[3]
- Activation of α-keto carboxylic acids and N-heterocyclic carboxylic acids for alkynylation to form ynediones and N-heterocyclic ynones, respectively.[4]
- Synthesis of N-heterocyclic ynones and ynediones, used to activate carboxylic acids
- Chlorination and halogenation
- Three-component [3+2] cycloadditions
- Reactions with organostannanes
- Synthesis of cyclopentenones
- Carbonylations, used as a carbonyl synthon
Packaging
Legal Information
accessory
signalword
Danger
Hazard Classifications
Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Eye Dam. 1 - Flam. Liq. 2 - Skin Corr. 1B - Water-react 1
supp_hazards
Storage Class
4.3 - Hazardous materials which set free flammable gases upon contact with water
wgk_germany
WGK 1
flash_point_f
51.8 °F - closed cup
flash_point_c
11.0 °C - closed cup
ppe
Faceshields, Gloves, Goggles
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service