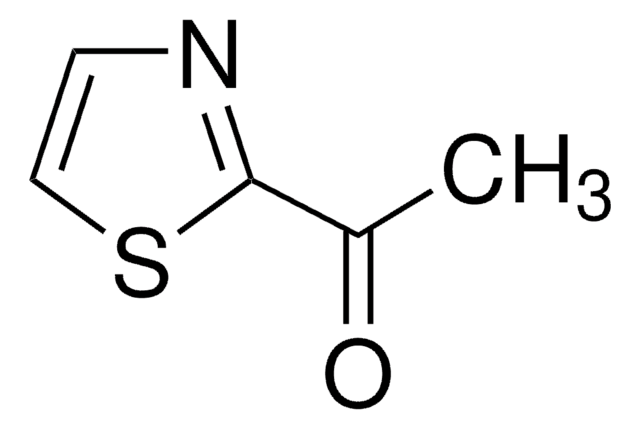
247359
2-Acetylpyrrole
ReagentPlus®, 99%
Synonym(s):
Methyl 2-pyrrolyl ketone
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C6H7NO
CAS Number:
Molecular Weight:
109.13
Beilstein/REAXYS Number:
1882
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
product line
ReagentPlus®
assay
99%
bp
220 °C (lit.)
mp
88-93 °C (lit.)
functional group
ketone
SMILES string
CC(=O)c1ccc[nH]1
InChI
1S/C6H7NO/c1-5(8)6-3-2-4-7-6/h2-4,7H,1H3
InChI key
IGJQUJNPMOYEJY-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
2-Acetylpyrrole undergoes alkylation reaction with alkyl iodide in benzene/solid KOH system in the presence of 18-crown-6 to yield the corresponding 1-alkyl derivative.
Application
2-Acetylpyrrole has been used in the synthesis of 2-acetyl-1-pyrroline.
Legal Information
ReagentPlus is a registered trademark of Merck KGaA, Darmstadt, Germany
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Cooked rice aroma and 2-acetyl-1-pyrroline.
Buttery RG, et al.
Journal of Agricultural and Food Chemistry, 31(4), 823-826 (1983)
Alkylation of 2-Acetylpyrrole and 1-alkyl-2-Acetylpyrroles Under Solid/Liquid Phase-Transfer Conditions.
Goldberg Y, et al.
Synthetic Communications, 21(4), 557-562 (1991)
Ana Filipa L O M Santos et al.
The journal of physical chemistry. A, 113(15), 3630-3638 (2009-03-27)
A combined experimental and computational study on the thermochemistry of 2- and 3-acetylpyrroles was performed. The enthalpies of combustion and sublimation were measured by static bomb combustion calorimetry and Knudsen effusion mass-loss technique, respectively, and the standard (p(o) = 0.1
Jin Ho Lee et al.
The Journal of organic chemistry, 78(3), 1283-1288 (2013-01-08)
A new synthetic route to indolizines with various substituents on the pyridine moiety was developed by utilizing a facile cycloaromatization of 2-acetylpyrrole derivatives. Without isolation, the resulting intermediates were allowed to react with various electrophiles to afford a range of
Wen-Yong Liu et al.
Journal of Asian natural products research, 5(3), 159-163 (2003-08-23)
Three pyrrole alkaloids were isolated from Bolbostemma paniculatum. Their structures were elucidated as 4-(2-formyl-5-methoxymethylpyrrol-1-yl)butyric acid methyl ester (1), 2-(2-formyl-5-methoxymethylpyrrol-1-yl)-3-phenylpropionic acid methyl ester (2) and alpha-methyl pyrrole ketone (3) by spectroscopic techniques. Among them, 1 and 2 are new compounds.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service