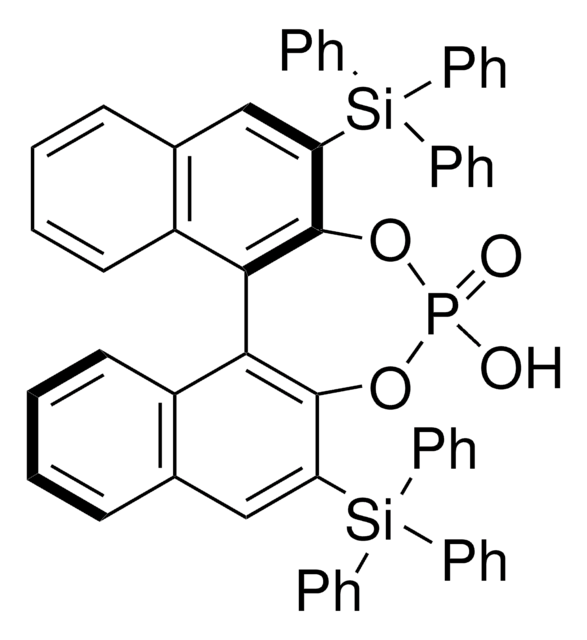
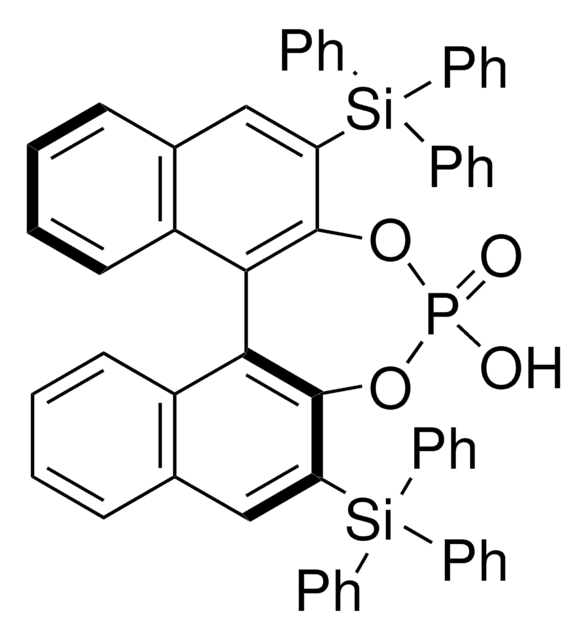
248932
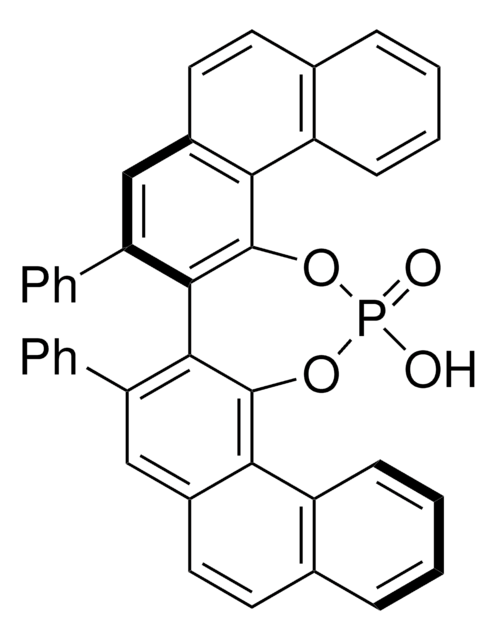
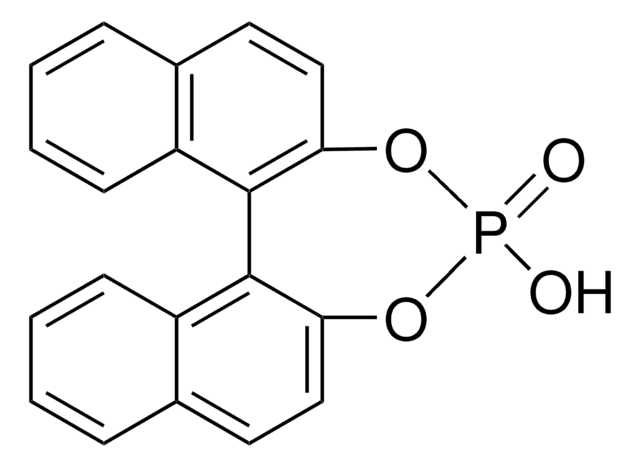
(R)-(−)-1,1′-Binaphthyl-2,2′-diyl hydrogenphosphate
≥98%
Synonym(s):
(R)-(−)-1,1′-Binaphthalene-2,2′-diyl hydrogen phosphate, (R)-4-Hydroxydinaphtho[2,1-d:1′,2′-f][1,3,2]dioxaphosphepin-4-oxide
About This Item
Recommended Products
Quality Level
assay
≥98%
form
solid
optical activity
[α]20/D −605°, c = 1.35 in methanol
functional group
phosphate
InChI
1S/C20H13O4P/c21-25(22)23-17-11-9-13-5-1-3-7-15(13)19(17)20-16-8-4-2-6-14(16)10-12-18(20)24-25/h1-12H,(H,21,22)
InChI key
JEHUZVBIUCAMRZ-UHFFFAOYSA-N
Application
- To facilitates the controlled ring-opening homopolymerization and copolymerization of ε-caprolactone (cyclic ester).
- As an additive along with a copper catalyst to achieve the highest catalytic activity in Sonogashira-type reactions.
signalword
Warning
hcodes
Hazard Classifications
Skin Irrit. 2
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
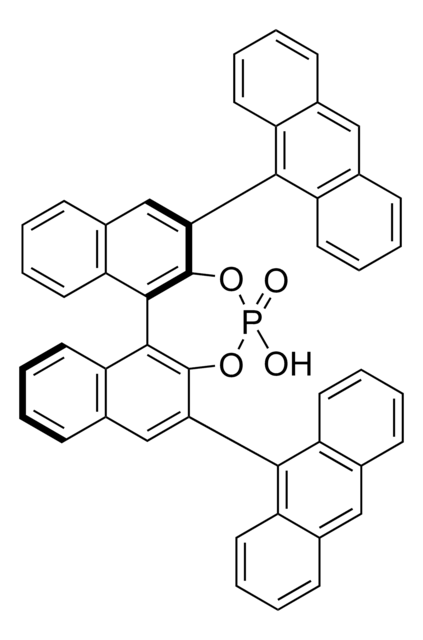
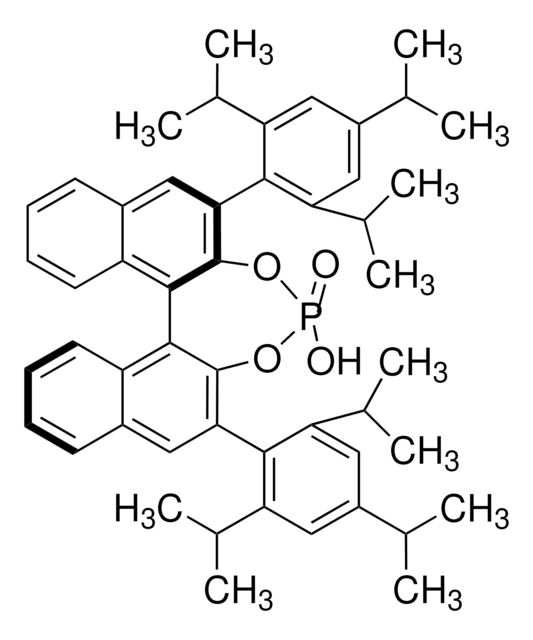
Customers Also Viewed
Articles
TRIP and TiPSY Chiral Phosphoric Acid Catalysts
Chromatograms
application for HPLCOur team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![(R)-3,3′-Bis[3,5-bis(trifluoromethyl)phenyl]-1,1′-binaphthyl-2,2′-diyl hydrogenphosphate 95%](/deepweb/assets/sigmaaldrich/product/structures/270/636/14dc9413-bcb4-478c-8e4d-3605317c13a5/640/14dc9413-bcb4-478c-8e4d-3605317c13a5.png)

![(11bR)-2,6-Di-9-phenanthrenyl-4-hydroxy-dinaphtho[2,1-d:1′,2′-f][1,3,2]dioxaphosphepin-4-oxide](/deepweb/assets/sigmaaldrich/product/structures/340/448/84d0af38-2286-4204-b2fa-7ce44f4b40bc/640/84d0af38-2286-4204-b2fa-7ce44f4b40bc.png)