391107
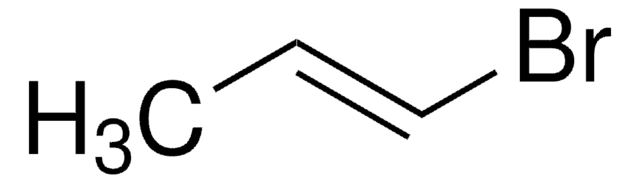
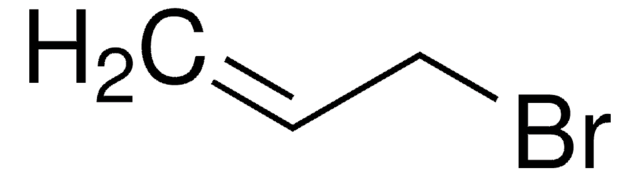
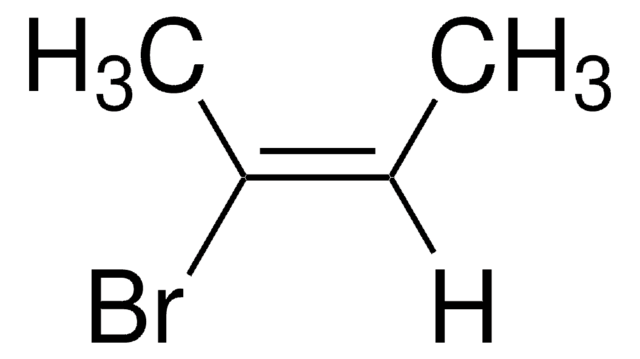
trans-1-Bromo-1-propene
contains copper as stabilizer, 99%
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
CH3CH=CHBr
CAS Number:
Molecular Weight:
120.98
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
99%
form
liquid
contains
copper as stabilizer
refractive index
n20/D 1.453 (lit.)
bp
64-65 °C (lit.)
density
1.408 g/mL at 25 °C (lit.)
functional group
alkyl halide
bromo
storage temp.
2-8°C
SMILES string
C\C=C\Br
InChI
1S/C3H5Br/c1-2-3-4/h2-3H,1H3/b3-2+
InChI key
NNQDMQVWOWCVEM-NSCUHMNNSA-N
Related Categories
General description
trans-1-Bromo-1-propene is an alkenyl halide. Product contains copper as stabilizer. Its synthesis from 1,2-dibromopropane has been reported by various researchers. Its IR spectra has been investigated.
Application
trans -1-Bromo-1-propene is suitable for the synthesis of (E)-hex-4-en-2-yn-1-ol. trans -1-Bromo-1-propene ((E)-1-bromo-1-propene) may be used in the synthesis of the following:
- (E)-1,3-dimethyl-3-(prop-1-enyl)indolin-2-one
- 3-alkenyl-Δ3-cephems
- (E)-1-(benzylthio)-1-propene
trans-1-Bromo-1-propene may be used as starting reagent in the synthesis of (+)-trans-isoalliin and (-)-trans-isoalliin.
accessory
Product No.
Description
Pricing
signalword
Danger
hcodes
Hazard Classifications
Eye Irrit. 2 - Flam. Liq. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
3 - Flammable liquids
wgk_germany
WGK 3
flash_point_f
5.0 °F - closed cup
flash_point_c
-15 °C - closed cup
ppe
Eyeshields, Faceshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Substituted Dienols from Palladium Catalyzed Coupling of Hydroaluminated Enynols with Aryl Iodides.
Crook KE, et al.
Letters in Organic Chemistry, 5(3), 158-164 (2008)
H Tanaka et al.
The Journal of organic chemistry, 66(2), 570-577 (2001-06-30)
Synthesis of 3-alkenyl-delta 3-cephems was performed successfully by cross-coupling 3-(trifluoromethylsulfonyloxy or chloro)-delta 3-cephem with alkenyl halides, e.g., vinyl bromide, trans-1-bromo-1-propene, and trans-beta-bromostyrene in an Al/cat.PbBr2/cat.NiBr2(bpy)/NMP (or DMF) system. Reduction of 3-(trifluoromethylsulfonyloxy)-delta 3-cephem into norcephalosporin was also achieved by a similar
Lasanthi Jayathilaka et al.
Journal of biomolecular techniques : JBT, 25(3), 67-76 (2014-09-05)
Naturally occurring (+)-trans-isoalliin, (R(C)R(S))-(+)-trans-S-1-propenyl-L-cysteine sulfoxide, is a major cysteine sulfoxide in onion. The importance of producing it synthetically to support further research is very well recognized. The (+)-trans-isoalliin is prepared by chemical synthesis and reversed-phase (RP)-HPLC. First, S-2-propenyl-L-cysteine (deoxyalliin) is
Structure Analysis of Poly (propylene-β-d oxide) by Proton Nuclear Magnetic Resonance Spectroscopy.
Oguni N, et al.
Macromolecules, 6(3), 459-465 (1973)
Facile synthesis of trans-S-1-propenyl-L-cysteine sulfoxide (isoalliin) in onions (Allium cepa).
Lee S, et al.
Bull. Korean Chem. Soc., 32, 319-320 (2011)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service