439320
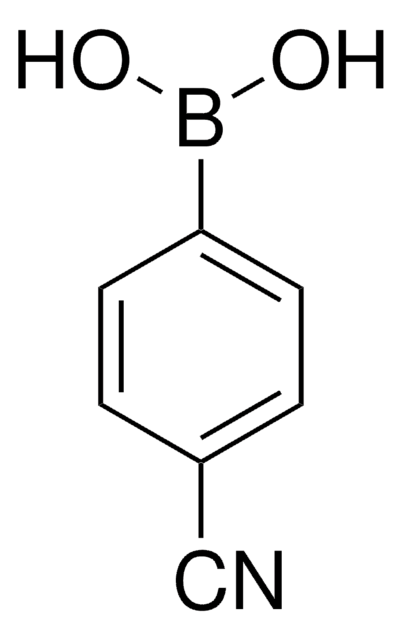
4-(Trifluoromethyl)phenylboronic acid
≥95.0%
Synonym(s):
α,α,α-Trifluoro-p-tolylboronic acid, 4-(Trifluoromethyl)benzeneboronic acid, [p-(Trifluoromethyl)phenyl]boronic acid
About This Item
Recommended Products
assay
≥95.0%
mp
245-250 °C (lit.)
functional group
fluoro
SMILES string
OB(O)c1ccc(cc1)C(F)(F)F
InChI
1S/C7H6BF3O2/c9-7(10,11)5-1-3-6(4-2-5)8(12)13/h1-4,12-13H
InChI key
ALMFIOZYDASRRC-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
- Site-selective Suzuki-Miyaura cross-coupling reactions.
- Palladium-catalyzed direct arylation reactions.
- Tandem-type Pd(II)-catalyzed oxidative Heck reaction and intramolecular C-H amidation sequence.
- Ruthenium catalyzed direct arylation.
- Ligand-free copper-catalyzed coupling reactions.
- Amination and conjugate addition reactions.
- Regioselective arylation and alkynylation by Suzuki-Miyaura and Sonogashira cross-coupling reactions.
- Rhodium-catalyzed asymmetric 1,4-addition reactions.
- Copper-catalyzed nitration reactions.
- Regioselective Suzuki-Miyaura coupling and tandem palladium-catalyzed intramolecular aminocarbonylation and annulation.
- Palladium catalyzed allylation reaction with allyl alcohols.
- N-Arylation of imidazoles and amines in the presence of copper-exchanged fluorapatite as a catalyst.
It can also be used as a reactant to prepare:
- Thiazole derivatives for printable electronics.
- Terphenyl benzimidazoles as tubulin polymerization inhibitors.
- Aryl ketones by cross-coupling reaction with acid chlorides.
Other Notes
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Oral
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service