754218
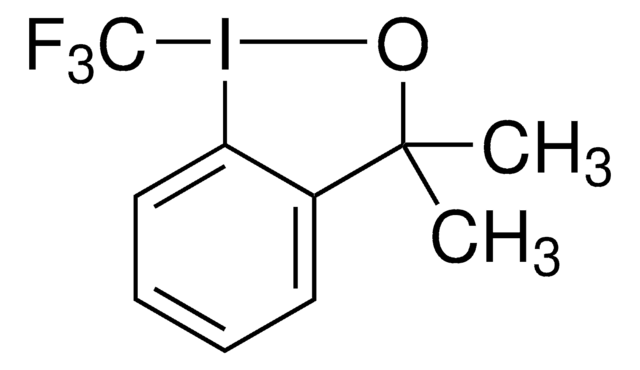
3,3-Dimethyl-1-(trifluoromethyl)-1,2-benziodoxole
98%
Synonym(s):
1,3-Dihydro-3,3-dimethyl-1-(trifluoromethyl)-1,2-benziodoxole, Togni’s Reagent
About This Item
Recommended Products
assay
98%
form
powder
mp
75-79 °C
storage temp.
2-8°C
SMILES string
CC1(C)O[I](c2ccccc12)C(F)(F)F
InChI
1S/C10H10F3IO/c1-9(2)7-5-3-4-6-8(7)14(15-9)10(11,12)13/h3-6H,1-2H3
InChI key
HVAPLSNCVYXFDQ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- Selective trifluoromethylation of 1,3-disubstituted arenes through iridium-catalyzed arene borylation and copper-catalyzed trifluoromethylation
- Copper-catalyzed trifluoromethylation of aryl- and alkenylboronic acids with electrophilic trifluoromethylating reagent
- Pd-catalyzed electrophilic ortho-trifluoromethylation of arenes using trifluoroacetic acid as a promotor
Used in the Preparation of
- Trifluoromethylimidoylethyl substituted heterocycles via bis(trifluoromethylsulfonyl)amine-catalyzed Rotter type reaction of heterocycles with nitriles in presence of trifluoromethylbenziodoxole
- Stereoselective synthesis of α-trifluoromethyl aldehydes via trimethylbenzylimidazolidinone and copper-catalyzed enantioselective α-trifluoromethylation of aldehydes with iodonium salts
Related product
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Flam. Sol. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
4.1B - Flammable solid hazardous materials
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Related Content
Research in the Togni group focuses on the development of new ligands and reagents. These two general directions thus impact the ability to construct molecules in more efficient or unprecedented ways.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service