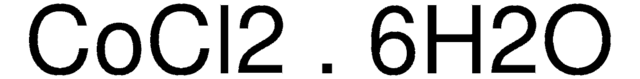769495
Cobalt(II) chloride hexahydrate
≥97%
Synonym(s):
Cobaltous chloride hexahydrate
About This Item
97.0-102.0% (KT)
Recommended Products
vapor pressure
40 mmHg ( 0 °C)
Quality Level
assay
≥97%
97.0-102.0% (KT)
form
solid
anion traces
nitrate (NO3-): ≤0.01%
sulfate (SO42-): ≤0.007%
cation traces
Fe: ≤0.005%
Ni: ≤0.15%
Pb: ≤0.002%
Zn: ≤0.05%
SMILES string
O.O.O.O.O.O.Cl[Co]Cl
InChI
1S/2ClH.Co.6H2O/h2*1H;;6*1H2/q;;+2;;;;;;/p-2
InChI key
GFHNAMRJFCEERV-UHFFFAOYSA-L
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- An additive to the electron transport layer (ETL) in perovskite solar cells to improve their performance, particularly by reducing energy losses and increasing the open-circuit voltage.
- A cobalt source for doping ZnO nanostructures. The incorporation of cobalt ions into the ZnO matrix is crucial for modifying its electronic and optical properties.
- A precursor to modify cobalt metal-organic framework (Co-MOF) derived carbon microspheres for application as anode materials in lithium-ion batteries.
Analysis Note
signalword
Danger
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Carc. 1B Inhalation - Eye Dam. 1 - Muta. 2 - Repr. 1B - Resp. Sens. 1 - Skin Sens. 1
Storage Class
6.1D - Non-combustible acute toxic Cat.3 / toxic hazardous materials or hazardous materials causing chronic effects
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service