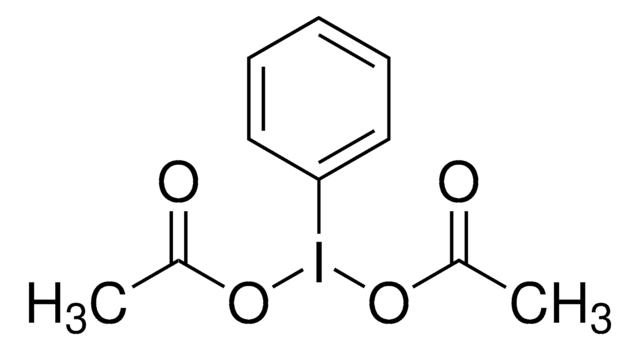
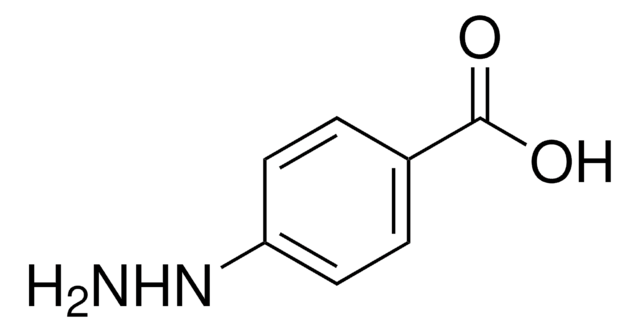
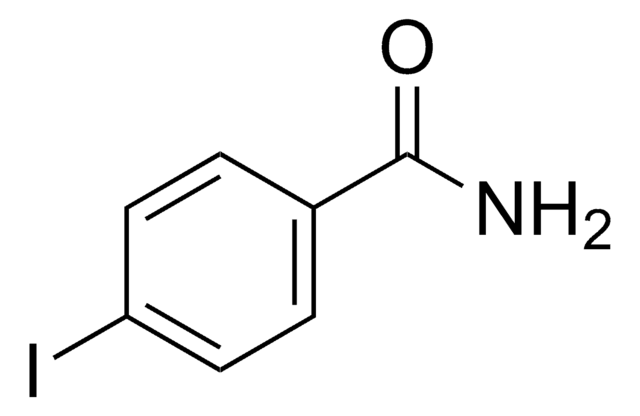
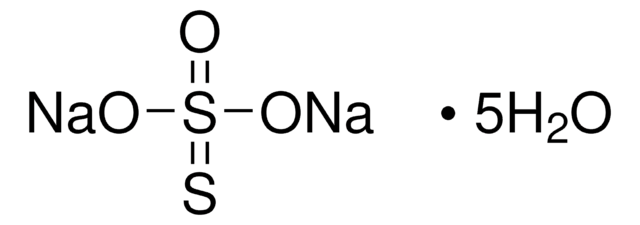About This Item
Recommended Products
Quality Level
assay
99%
form
solid
mp
140-144 °C (lit.)
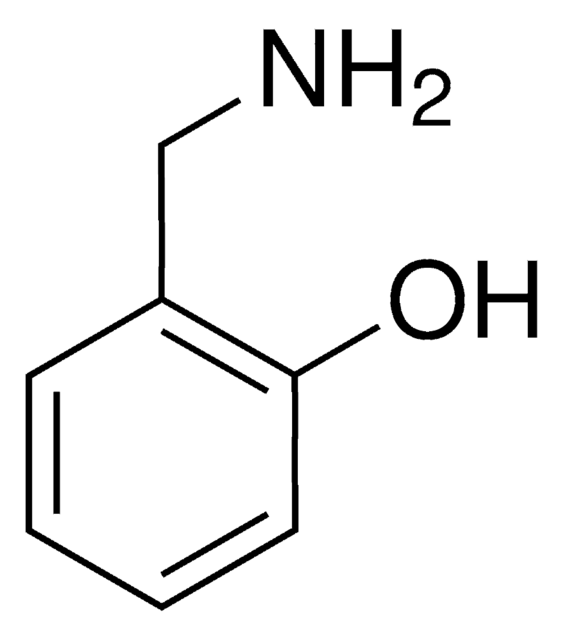
SMILES string
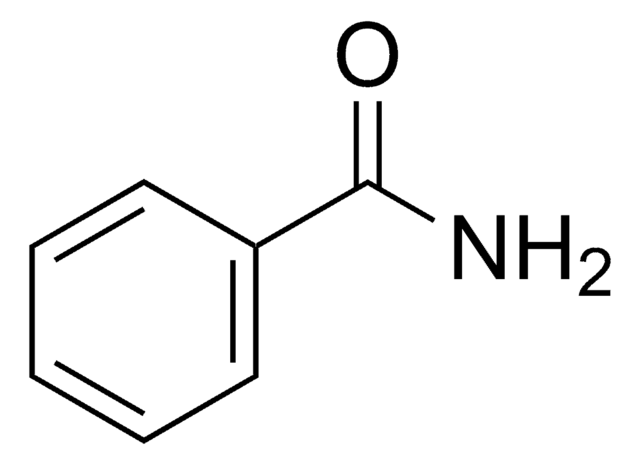
NC(=O)c1ccccc1O
InChI
1S/C7H7NO2/c8-7(10)5-3-1-2-4-6(5)9/h1-4,9H,(H2,8,10)
InChI key
SKZKKFZAGNVIMN-UHFFFAOYSA-N
Gene Information
human ... AR(367)
Looking for similar products? Visit Product Comparison Guide
General description
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 2
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service