All Photos(1)
About This Item
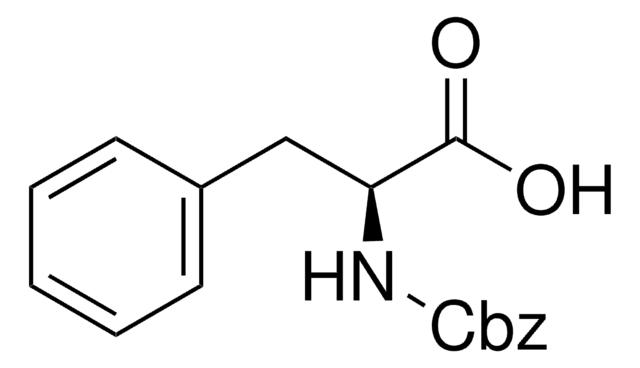
Linear Formula:
C6H5CH2OOCNHCH2COOH
CAS Number:
Molecular Weight:
209.20
Beilstein/REAXYS Number:
526877
EC Number:
MDL number:
UNSPSC Code:
12352209
eCl@ss:
32160406
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
99%
form
powder
reaction suitability
reaction type: solution phase peptide synthesis
color
beige
mp
118-122 °C (lit.)
application(s)
peptide synthesis
SMILES string
OC(=O)CNC(=O)OCc1ccccc1
InChI
1S/C10H11NO4/c12-9(13)6-11-10(14)15-7-8-4-2-1-3-5-8/h1-5H,6-7H2,(H,11,14)(H,12,13)
InChI key
CJUMAFVKTCBCJK-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Z-Gly-OH, also known as N-benzyloxycarbonylglycine, is an amino acid widely used in solution phase peptide synthesis.
Application
Z-Gly-OH is a versatile reagent that can be used to synthesize a variety of compounds such as:
- glycine-derived peptides like Z-Gly-DL-Ala-OBzl and Z-Gly-L-Ala-OBzl
- glycine N-substituted amides such as glycine-N-methylamide hydrochloride and glycine-N-isopropylamide hydrochloride
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Studies on Separation of Amino Acids and Related Compounds. V. A Racemization Test in Peptide Synthesis by the Use of an Amino Acid Analyzer
Bulletin of the Chemical Society of Japan, 44, 3391-3395 (1971)
Formation of peptide bonds in the coordination sphere of cobalt (III)
Journal of the American Chemical Society, 89, 6096-6103 (1967)
Duality of mechanism in the tetramethylfluoroformamidinium hexafluorophosphate-mediated synthesis of N-benzyloxycarbonylamino acid fluorides.
R Fiammengo et al.
The Journal of organic chemistry, 66(17), 5905-5910 (2001-08-21)
G K Scriba et al.
The Journal of pharmacy and pharmacology, 51(5), 549-553 (1999-07-20)
Glycine, which has weak anticonvulsant properties, has been shown to potentiate the activity of several antiepileptic drugs but not phenytoin. Recently, studies have shown that N-(benzyloxycarbonyl)glycine (Z-glycine) antagonized seizures more than glycine in addition to possessing activity in the maximal
D M Lambert et al.
Neuroreport, 5(7), 777-780 (1994-03-21)
Although glycine does not cross easily the blood-brain barrier, it exhibits at very high doses (10-40 mmol kg-1) a modest anticonvulsant activity. In this study, carbamate derivatives--N-benzyloxycarbonylglycine (Z-glycine) and N,tert-butoxycarbonylglycine (Boc-glycine)--have been compared with glycine. Z-glycine (1 mmol kg-1), but
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service