H36001
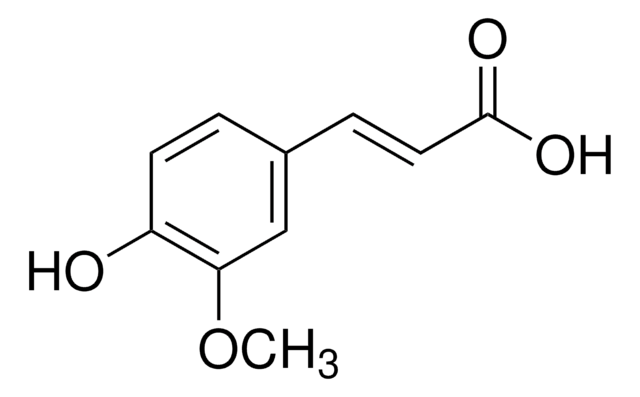
Vanillic acid
97%
Synonym(s):
4-Hydroxy-3-methoxybenzoic acid
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Linear Formula:
HOC6H3(OCH3)CO2H
CAS Number:
Molecular Weight:
168.15
Beilstein/REAXYS Number:
2208364
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
97%
mp
208-210 °C (lit.)
SMILES string
COc1cc(ccc1O)C(O)=O
InChI
1S/C8H8O4/c1-12-7-4-5(8(10)11)2-3-6(7)9/h2-4,9H,1H3,(H,10,11)
InChI key
WKOLLVMJNQIZCI-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Vanillic acid, also known as 4-Hydroxy-3-methoxybenzoic acid, is a phenolic molecule, commonly used as a building block in the synthesis of surfactants, flavorants, odorants, and plasticizers.
Application
- Vanillin Synthesis from Vanillic Acid: Research focused on engineering the activity and thermostability of a carboxylic acid reductase for converting vanillic acid to vanillin, providing insights into biotechnological applications for flavor and fragrance industries (Ren et al., 2024).
- Antioxidant Properties in Food Preservation: The antioxidant properties of vanillic acid were evaluated in a study on the preservation of postharvest quality and physicochemical properties of broccoli, suggesting its potential in extending the shelf life and nutritional quality of fresh produce (Kibar et al., 2024).
- Biological Synthesis of Vanillin: A comprehensive review discussed various biological methods for synthesizing vanillin from vanillic acid, emphasizing its application in enhancing natural flavor profiles in the food sector (Venkataraman et al., 2024).
Vanillic acid is used as a starting material in the synthesis of:
- aliphatic-aromatic polymers with good thermal stability and degradability
- novel polyesters via esterification and etherification reaction
Storage Class
11 - Combustible Solids
wgk_germany
WGK 1
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
An overview of therapeutic effects of vanillic acid
Neha S, et al.
Plant Archives, 20, 3053-3059 (2020)
Base metal iron catalyzed sustainable oxidation of vanillyl alcohol to vanillic acid in deep eutectic solvents and implementation of vanillic acid for fine-chemical synthesis
Rahul G, et al.
RSC Sustainability, 1, 1223-1232 (2023)
From Biomass to Bio-Based Polymers: Exploitation of Vanillic Acid for the Design of New Copolymers with Tunable Properties
Micaela V, et al.
Macromolecular Chemistry and Physics, 224, 2300001-2300001 (2023)
Synthesis and characterization of renewable polyesters based on vanillic acid
Silong Z, et al.
Journal of Applied Polymer Science, 137, 49189-49189 (2020)
Rafael Llorach et al.
Journal of proteome research, 8(11), 5060-5068 (2009-09-17)
Cocoa-phytochemicals have been related to the health-benefits of cocoa consumption. Metabolomics has been proposed as a powerful tool to characterize both the intake and the effects on the metabolism of dietary components. Human urine metabolome modifications after single cocoa intake
Protocols
Protocol for HPLC Analysis of Flavonoids on Ascentis® RP-Amide
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service