W359904
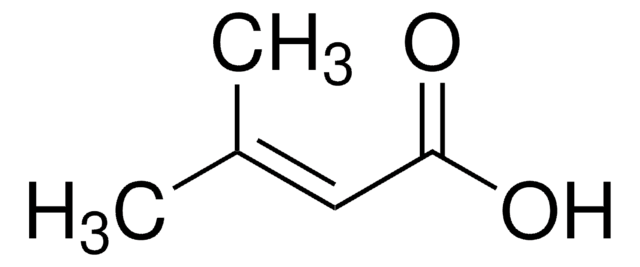
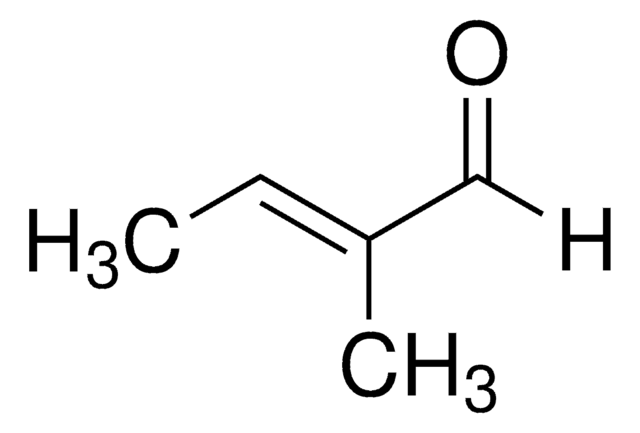
trans-2-Methyl-2-butenoic acid
≥99%, FG
Synonym(s):
Tiglic acid, trans-2,3-Dimethylacrylic acid, trans-2-Methyl-2-butenoic acid
About This Item
Fragrance grade
Halal
Kosher
meets purity specifications of JECFA
Recommended Products
biological source
synthetic
Quality Level
grade
FG
Fragrance grade
Halal
Kosher
agency
follows IFRA guidelines
meets purity specifications of JECFA
reg. compliance
EU Regulation 1223/2009
EU Regulation 1334/2008 & 178/2002
assay
≥99%
bp
95-96 °C/12 mmHg (lit.)
mp
61-64 °C (lit.)
density
0.969 g/mL at 25 °C (lit.)
application(s)
flavors and fragrances
documentation
see Safety & Documentation for available documents
food allergen
no known allergens
fragrance allergen
no known allergens
organoleptic
brown; spicy
SMILES string
C\C=C(/C)C(O)=O
InChI
1S/C5H8O2/c1-3-4(2)5(6)7/h3H,1-2H3,(H,6,7)/b4-3+
InChI key
UIERETOOQGIECD-ONEGZZNKSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
- A high molar extinction coefficient bisterpyridyl homoleptic Ru(II) complex with trans-2-methyl-2-butenoic acid functionality: potential dye for dye-sensitized solar cells.: This study explores the synthesis and application of a Ru(II) complex incorporating trans-2-methyl-2-butenoic acid as a dye for dye-sensitized solar cells. The high molar extinction coefficient and favorable photophysical properties make this compound a promising candidate for enhancing solar cell efficiency (Adeloye et al., 2012).
- Synthesis, photophysical and electrochemical properties of a mixed bipyridyl-phenanthrolyl ligand Ru(II) heteroleptic complex having trans-2-methyl-2-butenoic acid functionalities.: This research focuses on the synthesis and characterization of a Ru(II) heteroleptic complex with trans-2-methyl-2-butenoic acid functionalities. The study highlights the compound′s photophysical and electrochemical properties, demonstrating its potential for use in optoelectronic devices and catalytic applications (Adeloye, 2011).
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 2
flash_point_f
203.0 °F
flash_point_c
95 °C
ppe
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service