T4764
Δ9-Tetrahydrocannabinol solution
1.0 mg/mL in methanol, analytical standard, for drug analysis
Synonym(s):
Δ1-Tetrahydrocannabinol
About This Item
Recommended Products
grade
analytical standard, for drug analysis
Quality Level
drug control
Home Office Schedule 2; stupéfiant (France); kontrollierte Droge in Deutschland; (Spain); Decreto Lei 15/93: Tabela IIB (Portugal)
concentration
1.0 mg/mL in methanol
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
application(s)
pharmaceutical (small molecule)
format
single component solution
storage temp.
2-8°C
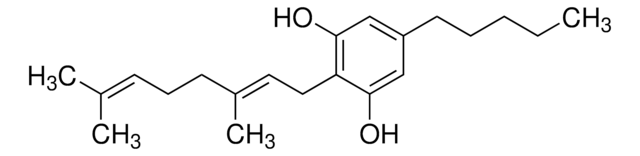
SMILES string
[H][C@@]12CCC(C)=C[C@@]1([H])c3c(O)cc(CCCCC)cc3OC2(C)C
InChI
1S/C21H30O2/c1-5-6-7-8-15-12-18(22)20-16-11-14(2)9-10-17(16)21(3,4)23-19(20)13-15/h11-13,16-17,22H,5-10H2,1-4H3/t16-,17-/m1/s1
InChI key
CYQFCXCEBYINGO-IAGOWNOFSA-N
Gene Information
human ... CNR1(1268) , CNR2(1269)
mouse ... Cnr1(12801)
rat ... Cnr1(25248)
Looking for similar products? Visit Product Comparison Guide
General description
Application
- Development and validation of a high-performance liquid chromatographic method coupled with UV detection to measure cannabidiol and ∆9-tetrahydrocannabinol in rat plasma samples following their extraction by protein precipitation and liquid-liquid extraction (LLE)
- Estimation of cannabidiol and ∆9-tetrahydrocannabinol extracted from human plasma samples by liquid-liquid extraction using ultra-performance liquid chromatography with triple quadrupole mass spectrometry
- Multi-residue analysis of Δ9-tetrahydrocannabinol (THC) and its two degradation products, 11-hydroxy-Δ9-tetrahydrocannabinol and 11-nor-9-carboxy-Δ9-tetrahydrocannabinol, in whole blood samples by QuEChERS extraction and gas chromatography (GC) coupled with tandem mass spectrometry (MS/MS)
- Optimization of an ultra-high performance supercritical fluid chromatographic (UHPSFC) method, combined with a tandem mass spectrometer (MS/MS) to determine ∆9-tetrahydrocannabinol, along with three of its major metabolites and four synthetic metabolites in wastewater samples following liquid-liquid extraction
- Development and validation of an ultra high-performance liquid chromatographic (UHPLC) method coupled to photodiode array and mass spectrometry detectors to determine 11 cannabinoids in various Cannabis Sativa samples
Other Notes
signalword
Danger
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Flam. Liq. 2 - STOT SE 1
target_organs
Eyes,Central nervous system
Storage Class
3 - Flammable liquids
wgk_germany
WGK 2
flash_point_f
49.5 °F - closed cup
flash_point_c
9.7 °C - closed cup
ppe
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service