Mesenchymal Stem Cell Culture Protocols
Isolation of Mesenchymal Stem Cells
Expansion of Human Mesenchymal Stem Cells
Differentiation of Human Mesenchymal Stem Cells
Frequently Asked Questions
Mesenchymal stem cells (MSCs) have the capacity for multi-lineage differentiation, giving rise to a variety of mesenchymal phenotypes such as osteoblasts (bone), adipocytes (fat), and chondrocytes (cartilage). Stem cell therapy holds immense promise of delivering the next generation of future medical breakthroughs. In this respect, multipotent progenitor cells, such as hMSCs, have attracted high clinical interest because of their ability to differentiate into various cell types and their immunoregulatory properties. Together, these features enable the allogeneic use of hMSCs and thus make them an attractive target for commercial therapeutic development. Below you will find step-by-step protocols used to isolate, expand and differentiate MSC’s properly in stem cell cultures.
Browse all Mesenchymal Stem Cell Products
Methods
Isolation of Mesenchymal Stem Cells
Mesenchymal stem cells have been isolated from a variety of tissues including human bone marrow, adipose tissue, umbilical cord and dental pulp. Below is a simple general protocol that can be used to derive MSCs from a variety of tissue sources.
Note: MSC populations will vary from donor to donor and might require further optimization.
- Obtain fresh tissue aspirates that are collected under aseptic conditions.
- Filter the cell suspension through a 70-mm filter mesh (CLS431751) to remove any cell clumps.
- Pellet the cells at 500g for 5 min in a benchtop centrifuge.
- Resuspend the cells and determine the yield and viability of cells by Trypan blue (T8154) exclusion
- Culture cells in T75 culture dishes in 10 mL of complete MSC medium (SCM015 or SCM045) at a density of 25 × 106 cells/mL. Incubate the plates at 37 °C with 5% CO2 in a humidified chamber without disturbing them.
- After 3 h, remove the non-adherent cells that accumulate on the surface of the dish by changing the medium and replacing with 10 mL fresh complete medium.
- After an additional 8 h of culture, replace the medium with 10 mL of fresh complete medium. Thereafter, repeat this step every 8 h for up to 72 h of initial culture.
- Cells can be frozen in MSC growth media plus 10% DMSO (D2650) at a density of 2X106 cells/vial.
Expansion of Human Mesenchymal Stem Cells
A. Preparation of Coated Plates
Tissue culture plastic- or glassware plates should be coated with 0.1% gelatin as follows:
- Add sufficient 0.1% gelatin solution (ES-006-B) to cover the entire surface of the cultureware plate. Use 10 mL volume for 10-cm plates and T75 flasks. Incubate for at least 30 minutes at room temperature.
- Just before use, aspirate the gelatin solution from the coated plate or flask.
B. Thawing of Mesenchymal Stem Cells
- Do not thaw the cells until the recommended medium and appropriately coated 0.1% gelatin plasticware and/or glassware are on hand.
- Remove the vial of Human Mesenchymal Stem Cells (SCC034, SCC038) from liquid nitrogen and incubate in a 37 °C water bath. Closely monitor until the cells are completely thawed. Maximum cell viability is dependent on the rapid and complete thawing of frozen cells.
IMPORTANT: Do not vortex the cells.
- As soon as the cells are completely thawed, disinfect the outside of the vial with 70% ethanol. Proceed immediately to the next step.
- In a laminar flow hood, use a 1 or 2 mL pipette to transfer the cells to a sterile 15 mL conical tube. Be careful not to introduce any bubbles during the transfer process.
- Using a 10 mL pipette, slowly add dropwise 9 mL of Mesenchymal Stem Cell Expansion Medium (SCM015 or SCM045) or a suitable alternative of choice, pre-warmed to 37 °C, to the 15 mL conical tube.
IMPORTANT: Do not add the entire volume of media all at once to the cells. This may result in decreased cell viability due to osmotic shock.
- Gently mix the cell suspension by slowly pipetting up and down twice. Be careful to not introduce any bubbles.
IMPORTANT: Do not vortex the cells.
- Centrifuge the tube at 300 x g for 2-3 minutes to pellet the cells.
- Decant as much of the supernatant as possible. Steps 5-8 are necessary to remove residual cryopreservative (DMSO).
- Resuspend the cells in a total volume of 10 mL of Mesenchymal Stem Cell Expansion Medium (SCM015 or SCM045) or a suitable alternative of choice, pre-warmed to 37 °C, containing freshly added 8 ng/mL FGF-2 (F0291).
- Plate the cell suspension onto a 10-cm tissue culture plate or a T75 tissue culture flask.
IMPORTANT: Seeding density should be 5,000-6,000 cells/cm2
- Maintain the cells at 37 °C in a humidified incubator equilibrated with 5% CO2.
- The next day, exchange the medium with fresh Mesenchymal Stem Cell Expansion Medium (pre-warmed to 37 °C) containing 8 ng/mL FGF-2*. Replace with fresh medium containing FGF-2 every two to three days thereafter.
- When the cells are approximately 80% confluent, they can be dissociated with Trypsin-EDTA (SM-2003-C) and passaged further or frozen for later use.
NOTE: Depending on seeding density and passage number (i.e. later passages), cells may take longer to reach 80% confluency.
C. Expansion of Mesenchymal Stem Cells
- Subculture cells once they have reached approximately 80% confluence and are actively proliferating.
IMPORTANT: Subculture cells before reaching 100% confluency.
- Carefully remove the medium from the 10-cm tissue culture plate containing the confluent layer of human mesenchymal stem cells. Apply 3-5 mL of Trypsin-EDTA Solution (SM-2003-C) and incubate in a 37 °C incubator for 3-5 minutes.
- Inspect the plate and ensure the complete detachment of cells by gently tapping the side of the plate with the palm of your hand.
- Add 5 mL Mesenchymal Stem Cell Expansion Medium (SCM015 or SCM045) to the plate.
- Gently rotate the plate to mix the cell suspension. Transfer the dissociated cells to a 15 mL conical tube.
- Centrifuge the tube at 300 x g for 3-5 minutes to pellet the cells.
- Discard the supernatant
- Apply 2 mL Mesenchymal Stem Cell Expansion Medium (pre-warmed to 37 °C) containing 8 ng/mL FGF-2 to the conical tube and resuspend the cells thoroughly.
IMPORTANT: Do not vortex the cells.
- Count the number of cells using a hemocytometer.
- Plate the cells at a density of 5,000-6,000 cells per cm2 into the appropriate flasks, plates, or wells in Mesenchymal Stem Cell Expansion Medium containing 8 ng/mL FGF-2.
- Cells can be frozen in MSC growth media plus 10% DMSO (D2650) at a density of 2X106 cells/vial.
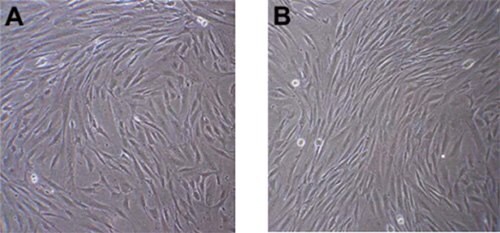
Figure 1.Phase contrast images of Human Mesenchymal Stem Cells one (A) and two (B) days after thawing. Note spindle shaped, fibroblast-like morphology. Right before passaging, cells should be ~80% confluent.

Figure 2. ICC staining of cultured human bone marrow-derived mesenchymal stem cells with antibodies provided in the Human Mesenchymal Stem Cell Characterization Kit. Human Mesenchymal Stem Cell express H-CAM (CD44) (A, CBL154; 1/500 dilution), THY-1 (CD90) (B, CBL415: 1/500 dilution) and STRO-1 (C, MAB4315: 1/500 dilution). Nuclei of the cells were visualized with DAPI (blue). Expression of hematopoietic stem cell markers, CD19 (MAB1794) and CD14 (MAB1219) and endothelial marker, CD146 (MAB16985) were not observed in human mesenchymal stem cells (data not shown).
Differentiation of Human Mesenchymal Stem Cells
Osteogenesis of Mesenchymal Stem Cells
Using a human fibronectin, 20 µg/mL (FC010) or a 0.1% gelatin (ES-006-B) coated 48 well tissue culture plate seed 20K cells/well in 0.5 mL normal MSC growth media (SCM015 or SCM045). After an overnight incubation replace the culture media with OsteoMAX-XF™ Differentiation Media (SCM121). Replace media every 2-3 days for a total of 14-21 days. Alizarin-Red (TMS-008) stained osteocytes will start forming around day 7 with maximum expression around day 14.
Adipogenesis of Mesenchymal Stem Cells
Using 24 well tissue culture plate seed 60K cells/well in 1 mL normal MSC growth media (SCM015 or SCM045). After an overnight incubation replace the culture media with AdipoMAX™ Differentiation Media (SCM122). Replace media every 2-3 days for a total of 14-21 days. Oil-Red-O (O1391) stained adipocytes will start forming around day 14 with maximum expression around day 21.
Chondrogenesis of Mesenchymal Stem Cells
Mesenchymal stem cells can be induced to differentiate into chondrocytes using three-dimensional micromass cultures of the cells in aggregates.
- Resuspend 2.5 x 105 MSCs in 5 mL pre-warmed ChondroMAX™ Differentiation Media (SCM123).
- Centrifuge the cells at 200g for 5 minutes.
- Remove the media and resuspend the cells with 0.5 mL pre-warmed ChondroMAX™ Differentiation Media.
- Centrifuge the cells at 200g for 5 minutes. Do not remove the media.
- Loosen the cap of the tube to allow gas exchange and incubate upright at 37 °C and 5% CO2.
- After 1 - 2 days the cell pellet will form a round ball approximately 1 - 2 mm in diameter. Carefully change medium every other day without damaging micromass pellet of cells. After 21 days micromass cultures can be fixed, sectioned and stained with Safranin-O (TMS-009), Alcian-Blue (TMS-010) or antibodies to identify chondrocytes.
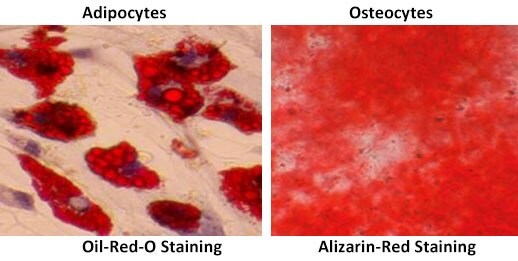

Figure 3.Staining of differentiated human mesenchymal stem cell cultures with Oil-Red-O (Adipocytes), Alizarin-Red (Osteocytes), Safranin-O (Chondrocytes) and Alcian-Blue (Chondrocytes).
Frequently Asked Questions
- How long do MSCs expand in culture? It is recommended to start with low passage number cells (≤P2) because MSCs rarely expand past passage 5-10 (10 population doublings).
- My cells are not proliferating anymore, what should I do? It is recommended to start with a new fresh vial of low passage cells.
- What is the typical morphology of multipotent MSCs? MSCs should have a spindle shaped/fibroblastic–like morphology. Cells become larger and less fibroblastic when reaching senescence.
- What markers can be used to characterize MSCs? MSCs uniformly express the integrin CD29, matrix receptors CD44, CD105 and stromal cell associated markers CD73, CD90, and CD166. The cells are negative for hematopoietic lineage markers CD14, CD31, CD34, and CD45.
- How do I know when to split my MSCs? Split cells when they are no more than 80% confluent.
- What is the correct seeding density to use when passaging MSCs? We recommend seeding new plates with cells at 5000 cells/cm2
- Can I use FBS to culture MSC’s? Media containing low concentrations of FBS (<1% FBS) can be used to expand MSCs. However, due to its undefined nature, each lot of FBS must be screened to support MSC growth. We recommend PLTMax® Human Platelet Lysate (SCM141) or Xeno-Free MSC media (SCM045) which do not require such pre-screening.
To continue reading please sign in or create an account.
Don't Have An Account?