Determination of Baicalin, Chlorogenic acid and Forsythin in Shuanghuanglian Oral Liquid acc. to Chinese Pharmacopeia
Dean Duan, Merck R&D
APAC Lab Shanghai, China
Abstract
In this application, Shuanghuang Lian oral liquid, a well-known traditional Chinese medicine (TCM), was prepared following Chinese Pharmacopoeia 2020 and analyzed on a Discovery® HS C18 HPLC column for the content of three key compounds, baicalin, chlorogenic acid, and forsythin (phillyrin). For baicalin the linear range was 0.5 – 200 µg/mL. LOD and LOQ of baicalin is 0.07 µg/mL and 0.23 µg/mL separately. For chlorogenic acid the linear range is 0.1 – 50 µg/mL. LOD and LOQ of baicalin was 0.11 µg/mL and 0.33 µg/mL separately. For forsythin the linear range was 0.2 – 60 µg/mL. LOD and LOQ of baicalin was 0.02 µg/mL and 0.07 µg/mL separately. The SPE cleanup for the forsythin sample showed good recoveries of 88%. The method met the requirements of the Chinese Pharmacopeia 2020. The Discovery® HS C18 HPLC column can be used to determine the baicalin, chlorogenic acid, and forsythin content of Shuanghuang Lian oral liquid.
Section Overview
Introduction
Shuang-Huang Lian oral liquid is a Chinese patent medicine. It is composed of honeysuckle, scutellaria, and forsythia. The determination of baicalin, chlorogenic acid, and forsythin (phillyrin) as key compounds in the oral liquid by HPLC is listed in the Chinese Pharmacopeia 2020.1 Here we describe three dedicated analytical methods using a Discovery® HS C18 HPLC column according to the pharmacopeia monograph.

Experimental
HPLC conditions and sample preparation applied for the three analytes of interest are shown in Tables 1–3. For baicalin and chlorogenic acid, a dilution and filtration were applied, for forsythin, a clean-up by SPE (interference removal, chemical filtration) using a neutral alumina as adsorbent was used.
HPLC Parameters & Samples- Baicalin | |
|---|---|
Column: | Discovery® HS C18 250x4.6 mm, 5 µm (568523-U) |
Mobile phase: | [A] water; [B] methanol; [C] glacial acetic acid A:B:C = 50:50:1 |
Gradient: | isocratic |
Flow rate: | 1.0 mL/min |
Pressure drop: | 2700 psi |
Colum temp.: | 25°C |
Detector: | UV at 274 nm |
Injection Volume: | 5 µL |
Samples | |
Standard for System Suitability: | Dissolve appropriate amount of baicalin in 50% aqueous methanol solution to obtain 1 mg/mL stock solution, then dilute with 50% aqueous methanol solution to obtain 50 µg/mL standard solution |
Sample preparation: | Accurately measure 1 mL of Shuanghuang Lian oral liquid in a 50 mL volumetric flask with a stopper, add appropriate amount of 50% aqueous methanol, sonicate for 20 minutes, place it at room temperature, add 50% aqueous methanol solution to the tick mark of volumetric flask, mix well, filter (0.45 µm), stand for HPLC analysis. |
HPLC Parameters & Samples- Chlorogenic acid | |
|---|---|
Column: | Discovery® HS C18 250x4.6 mm, 5 µm (568523-U) |
Mobile phase: | [A] Water; [B] methanol; [C] glacial acetic acid. |
Gradient: | isocratic |
Flow rate: | 1.0 mL/min |
Pressure drop: | 2210 psi |
Colum temp.: | 25°C |
Detector: | UV, 324nm |
Injection: | 10 µL |
Samples | |
Standard for System Suitability: | Dissolve an appropriate amount of chlorogenic acid in water to obtain 1 mg/mL of chlorogenic acid stock solution, then dilute with water to obtain 20 µg/mL standard solution |
Sample preparation: | Accurately transfer 2 mL of Shuanghuang Lian oral liquid into a 50-mL brown volumetric flask, add water to required level, mix well, filter (0.45 µm), stand for HPLC analysis. |
HPLC Parameters & Samples - Forsythin | |
|---|---|
Column: | Discovery® HS C18 250x4.6 mm, 5 µm (568523-U) |
Mobile phase: | [A] Water; [B] acetonitrile. |
Gradient: | isocratic |
Flow rate: | 1.0 mL/min |
Pressure drop: | 1660 psi |
Colum temp.: | 25°C |
Detector: | UV at 278nm |
Injection Volume: | 10 µL |
Samples | |
Standard for System Suitability: | Dissolve an appropriate amount of forsythin (phillyrin) to 50% aqueous methanol solution to obtain 1 mg/mL of forsythin stock solution, then dilute with 50% aqueous methanol solution to obtain 30 µg/mL standard solution. |
Sample preparation: | SPE cleanup (Table 4): Accurately transfer 1 mL of Shuanghuang Lian oral liquid onto a neutral alumina SPE tube. Elute with 40 mL of 70% ethanol, collect the eluent, dry down under nitrogen at 45 ℃, add 5 mL of 50% methanol aqueous solution to dissolve the residue, filter with 0.45 µm filter membrane, stand for HPLC analysis. |
Sample Preparation by SPE for Forsythin Determination in Oral Liquid
For the determination of forsythin, an additional cleanup by SPE was applied.
SPE Sample Preparation | |
|---|---|
SPE tube/cartridge: | Supelclean™ LC-Alumina-N SPE Tube, 2 g / 6 mL (57087) |
Conditioning: | 5 mL of 70% aqueous ethanol solution |
Sample addition: | 1 mL Shuanghuang Lian oral liquid |
Washing: | none |
Elution: | 40 mL of 70% ethanol aqueous solution |
Elution post-treatment: | Evaporate to dryness under nitrogen flow at 45 ℃, add 5 mL of 50% methanol aqueous solution to dissolve the residue, and filter with 0.45 µm filter membrane |
Standards Preparation for Calibration
The calibration standards were prepared according to these procedures:
Diluent: Add 50 mL of methanol and 50 mL of water into a measuring cylinder. Mix well to prepare 50% aqueous methanol solution as diluent.
For Baicalin:
- Weigh ~10 mg of baicalin reference material into a 10 mL volumetric flask.
- Add ~8 mL of diluent and sonicate for 5 mins.
- Top-up to mark with diluent and mix well to prepare 1 mg/mL of baicalin stock solution.
- Dilute to 0.1, 0.2, 0.5, 1. 2, 5, 10, 50, 100 and 200 µg/mL with diluent.
For Chlorogenic acid:
- Weigh ~10 mg of chlorogenic acid reference material into a 10 mL volumetric flask.
- Add ~8 mL of diluent and sonicate for 5 mins.
- Top-up to mark with diluent and mix well to prepare 1 mg/mL of chlorogenic acid stock solution.
- Dilute to 0.1, 0.2, 0.5, 1. 2, 5, 10, 20, 40 and 50 µg/mL with diluent.
For Forsythin:
- Weigh ~10 mg of forsythin reference material into a 10 mL volumetric flask.
- Add ~8 mL of diluent and sonicate for 5 mins.
- Top-up to mark with diluent and mix well to prepare 1 mg/mL of forsythin stock solution.
- Dilute to 0.2, 0.5, 1. 2, 5, 10, 30, 40 and 60 µg/mL with diluent.
Method Suitability / System Suitability Criteria
Acceptance Criteria for Standard Solutions:
- Theoretical plate number calculated for baicalin peak: NLT 1500
- Theoretical plate number calculated for chlorogenic acid peak: NLT 6000
- Theoretical plate number calculated for forsythin peak: NLT 6000
Results & Discussion
The chromatographic results for the three compounds under investigation are displayed in Figures 1–5. The chromatographic data of the separately injected standard solutions is summarized in Table 5.
As an example of the calibration curves, the one for baicalin is displayed in Figure 6.
The methods displayed good reproducibility with RSDs of 0.98%, 0.27%, and 0.98% (Table 6), as well as good linearity and sensitivity (Table 7). In summary, the results for the three analytes determined are:
Baicalin:
- The theoretical plates is 10377 (>1500). It meets the requirements of the Chinese Pharmacopeia 2020.
- The Limit of Detection (LOD) for baicalin in Shuanghuang Lian oral liquid is 3.72 µg/mL, and the Limit of Quantification (LOQ) is 11.27 µg/mL. The range from 0.5 to 200 µg/mL is linear with an R2 of 0.9999.
Chlorogenic acid:
- The theoretical plates is 13128 (>6000). It meets the requirements of the Chinese Pharmacopeia 2020.
- The LOD for chlorogenic acid in Shuanghuang Lian oral liquid is 2.72 µg/mL, and the LOQ is 8.24 µg/mL. The range from 0.1 to 50 µg/mL is linear, with the R2 being 0.9982.
Forsythin:
- The theoretical plates is 8258 (>6000). It meets the requirements of the Chinese Pharmacopeia 2020.
- The LOD for forsythin in Shuanghuang Lian oral liquid is 0.11 µg/mL, and the LOQ is 0.33 µg/mL. The range from 0.2 to 60 µg/mL is linear, with the R2 being 0.9939.
- The average SPE recovery (n = 3) for forsythin was assessed using an oral liquid sample spiked at 75 µg/mL and was determined to be 88% (Table 8).
- The used oral liquid showed a forsythin content below LOD (Figure 4). The chromatogram for a sample spiked at 75 µg/mL is shown in Figure 5.
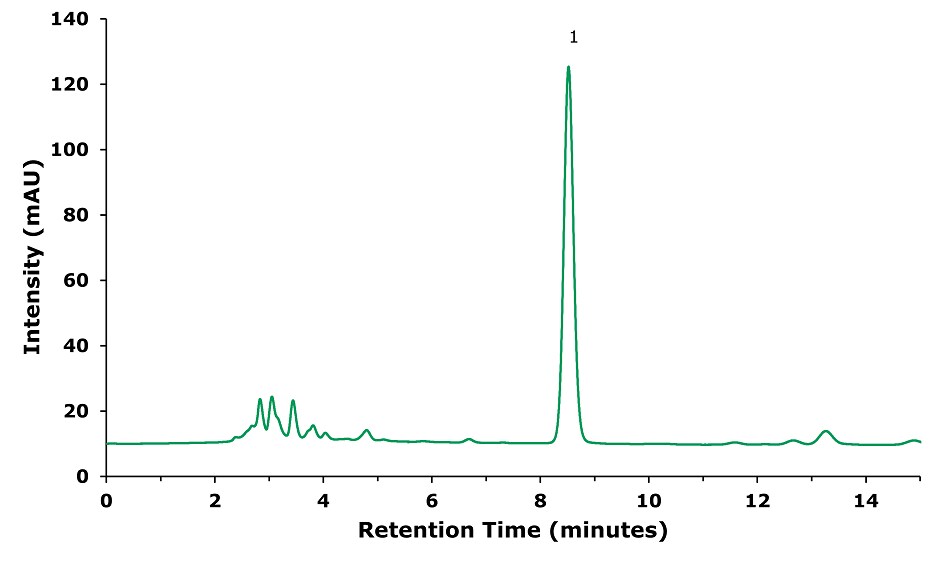
Figure 1.Sample Solution- Sample treated following procedure in baicalin (1) sample preparation part (Table 1).

Figure 2.Sample Solution- Sample treated following procedure in chlorogenic acid (1) sample preparation part (Table 2).

Figure 3.Chromatogram of forsythin (1) standard solution 30 µg/mL.

Figure 4.Sample Solution - Sample treated following the procedure in forsythin sample preparation part (Table 3). The used oral liquid showed forsythin content below LOD.

Figure 5.Oral liquid sample spiked with forsythin (1) at 75 µg/mL - Sample treated following the procedure in forsythin sample preparation part (Table 3).
Compound | Retention Time (min) | Theoretical Plates (Determined) | Theoretical Plates (Acceptance Criteria Ch P) | Tailing Factor |
|---|---|---|---|---|
Baicalin | 8.53 | 10377 | 1500 | 1.07 |
Chlorogenic acid | 11.79 | 13128 | 6000 | 1.04 |
Forsythin | 12.96 | 8258 | 6000 | 1.00 |
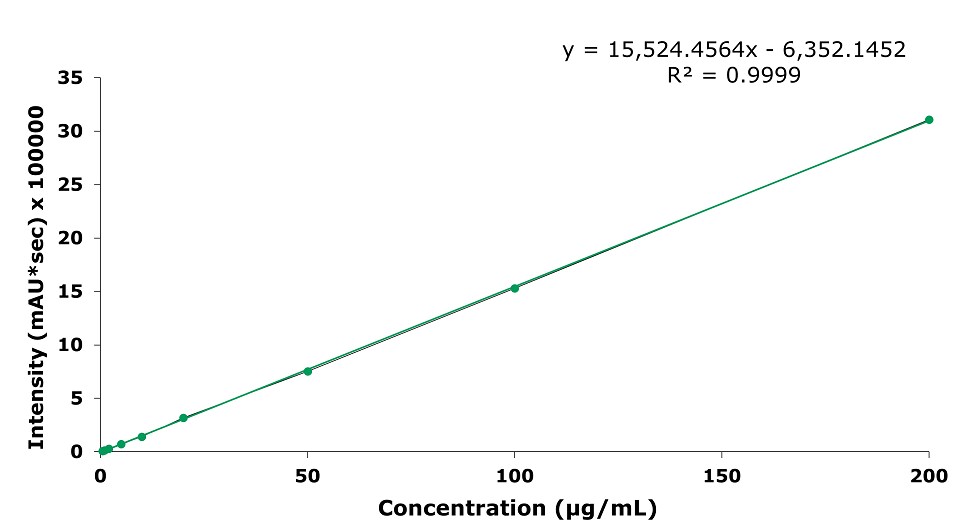
Figure 6.Calibration curve for baicalin.
Injection number | Baicalin, 50 µg/mL | Chlorogenic acid, 20 µg/mL | Forsythin, 30 µg/mL |
|---|---|---|---|
Injection 1 | 752679 | 470941 | 752679 |
Injection 2 | 758358 | 469506 | 758358 |
Injection 3 | 769377 | 472235 | 769377 |
Injection 4 | 770246 | 472652 | 770246 |
Injection 5 | 764743 | 471883 | 764743 |
Mean | 763081 | 471443 | 763081 |
Standard Deviation | 7487 | 1254 | 7487 |
RSD (%) | 0.98 | 0.27 | 0.98 |
Compound | Standards Calibration Range (µg/mL) | No. of Calibrators | R2 | Sample Preparation Dilution Factor | Oral Liquid LOD (µg/mL) | Oral Liquid LOQ (µg/mL) |
|---|---|---|---|---|---|---|
Baicalin | 0.5 -200 | 9 | 0.9999 | 1:50 | 3.72 | 11.27 |
Chlorogenic acid | 0.1 -50 | 10 | 0.9982 | 1:25 | 2.72 | 8.24 |
Forsythin | 0.2 -60 | 9 | 0.9939 | 1:5 | 0.11 | 0.33 |
Sample No. | Recovery (%) |
|---|---|
sample 1 | 90.1 |
sample 2 | 85.7 |
sample 3 | 88.1 |
Mean | 88.0 |
Standard Deviation | 2.0 |
RSD (%) | 2.5 |
Conclusion
The three HPLC methods using the Discovery® HS C18 column met the system suitability criteria listed in the Chinese Pharmacopeia monograph. The column is therefore suitable for the determination of baicalin, chlorogenic acid and forsythin in Shuanghuang Lian oral liquid. For the determination of forsythin, the used Supelclean™ LC-Alumina-N SPE Tube for clean-up of the oral liquid sample provided an average recovery (n=3) of 88%, which is suitable for the method.
See more applications on Pharmaceutical Analysis & Quality Control.
References
To continue reading please sign in or create an account.
Don't Have An Account?