PHR1817
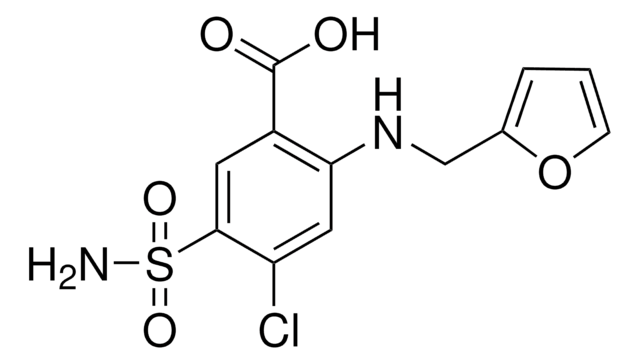
Furosemide Related Compound A
Pharmaceutical Secondary Standard; Certified Reference Material
別名:
2-Chloro-4-furfurylamino-5-sulfamoylbenzoic acid, 2-CHLORO-4-N-FURFURYLAMINO -5-SULFAMOYLBENZOIC ACID
About This Item
おすすめの製品
グレード
certified reference material
pharmaceutical secondary standard
品質水準
認証
traceable to Ph. Eur. F0702000
traceable to USP 1287020
CofA
current certificate can be downloaded
包装
pkg of 100 mg
mp
197 °C (dec.) (lit.)
アプリケーション
pharmaceutical
フォーマット
neat
保管温度
2-30°C
SMILES記法
OC(C1=C(Cl)C=C(NCC2=CC=CO2)C(S(N)(=O)=O)=C1)=O
InChI
1S/C12H11ClN2O5S/c13-9-5-10(15-6-7-2-1-3-20-7)11(21(14,18)19)4-8(9)12(16)17/h1-5,15H,6H2,(H,16,17)(H2,14,18,19)
InChI Key
UXOOVYKVEXGCSH-UHFFFAOYSA-N
類似した製品をお探しですか? 訪問 製品比較ガイド
詳細
アプリケーション
- Furosemide Impurity A for Quality Control: Furosemide Related Compound A serves as a crucial standard in pharmaceutical quality control, ensuring the purity and safety of furosemide as an active pharmaceutical ingredient. This impurity standard is essential for regulatory compliance and batch-to-batch consistency in drug manufacturing (Abasilim et al., 2023).
- Research on Drug Resistance: Furosemide Related Compound A is utilized in genomic research to identify potential drug resistance mechanisms. This research aids in understanding how bacterial pathogens develop resistance to drugs like clarithromycin and rifampicin, which is crucial for developing new therapeutic strategies (Mofidifar et al., 2024).
- Nanotechnology in Drug Delivery: High-purity Furosemide Related Compound A is used in the preparation and characterization of novel drug delivery systems, such as nanoparticles designed to enhance the bioavailability and efficacy of pharmaceutical compounds. This research highlights its application in improving drug formulations and targeting specific disease sites (Zeng et al., 2024).
- Impact on Bone Health: Research involving Furosemide Related Compound A explores its effects on bone health, particularly in how it influences vitamin D metabolism and bone density. This is vital for developing treatments that mitigate bone density loss associated with long-term use of diuretics like furosemide (Melanie KF et al., 2024).
- Nutritional Studies in Cancer Therapy: Furosemide Related Compound A is also examined for its role in nutritional interventions in cancer therapy, particularly its impact on the nutritional status of patients undergoing surgery for colorectal cancer. These studies are crucial for understanding how nutritional support can enhance recovery and treatment outcomes in cancer patients (Ural et al., 2024).
アナリシスノート
その他情報
脚注
おすすめ製品
保管分類コード
11 - Combustible Solids
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
適用法令
試験研究用途を考慮した関連法令を主に挙げております。化学物質以外については、一部の情報のみ提供しています。 製品を安全かつ合法的に使用することは、使用者の義務です。最新情報により修正される場合があります。WEBの反映には時間を要することがあるため、適宜SDSをご参照ください。
Jan Code
PHR1817-100MG-PW:
PHR1817-100MG:
最新バージョンのいずれかを選択してください:
この製品を見ている人はこちらもチェック
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)