추천 제품
일반 설명
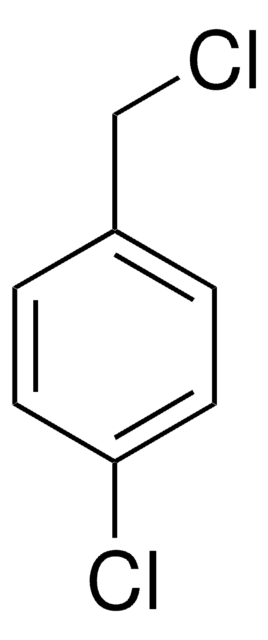
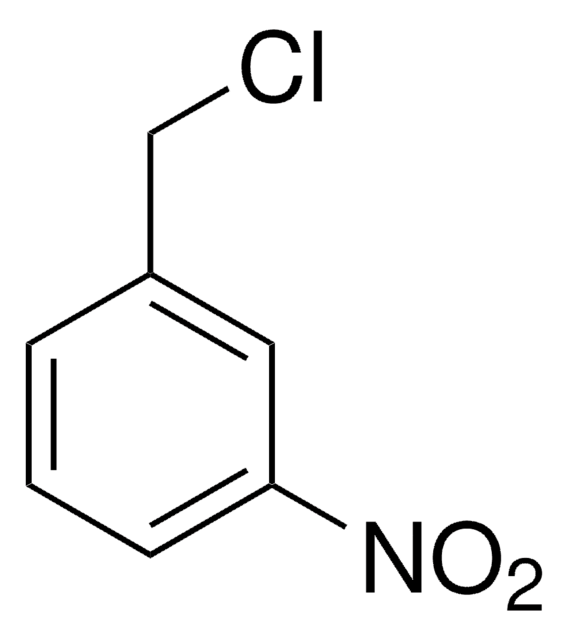
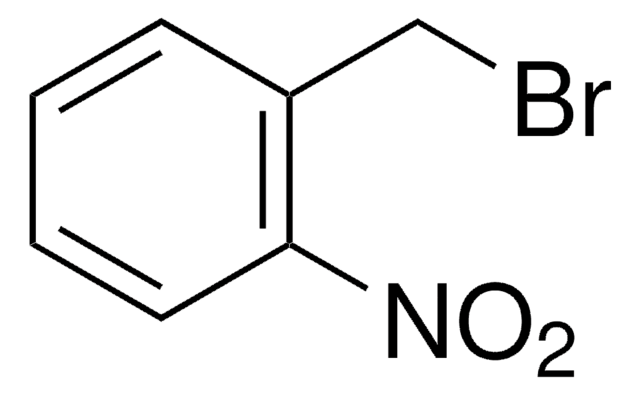
4-Nitrobenzyl chloride acts as substrate for glutathione S-transferase(GST) in determination of GST in Chinese fetal liver. It undergoes reduction by NADPH to yield 4-nitrotoluene.
애플리케이션
4-Nitrobenzyl chloride was used to prepare unsymmetrically N,N′-bis(substituted) 4,13-diaza-18-crown-6-ether derivatives.
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Oral - Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
이미 열람한 고객
P Jemth et al.
The Journal of biological chemistry, 275(12), 8618-8624 (2000-03-18)
Ser(11) in rat glutathione transferase T2-2 is important for stabilization of the reactive enzyme-bound glutathione thiolate in the reaction with 1-menaphthyl sulfate. The S11A mutation increased the pK(a) value for the pH dependence of the rate constant for pre-steady-state product
Tetrahedron Letters, 34, 95-95 (1993)
S B Lei et al.
Zhongguo yao li xue bao = Acta pharmacologica Sinica, 11(5), 389-391 (1990-09-01)
Subcellular fractions were isolated from Chinese fetal liver at 4-8 months of age for the determination of glutathione S-transferase (GST). Using 1-chloro-2,4-dinitrobenzene (CDNB) as substrate, GST activity was found to be 66 +/- 34 nmol/(min.mg protein), mainly in the cytosol.
S N Moreno et al.
The Journal of biological chemistry, 261(17), 7811-7815 (1986-06-15)
The o-, m-, and p-nitrobenzyl chlorides are reduced aerobically and anaerobically by NADPH and rat hepatic microsomes. Under aerobic conditions, these nitro anion radicals reduce oxygen to superoxide as demonstrated by oxygen consumption and spin trapping of superoxide with 5,5-dimethyl-1-pyrroline
P Jemth et al.
Biochemistry, 38(31), 9982-9991 (1999-08-06)
The reaction mechanism of rat glutathione transferase T2-2 has been studied using pre-steady-state and steady-state kinetics. Several parts of the catalytic cycle including binding of substrates, product formation, and product release were investigated. Under saturating conditions, a two-step product release
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.