모든 사진(2)
About This Item
Linear Formula:
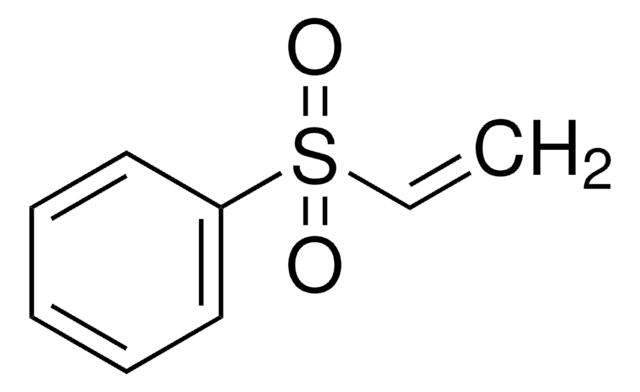
C6H5SOCH=CH2
CAS Number:
Molecular Weight:
152.21
Beilstein:
2039218
EC Number:
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
분석
95%
형태
liquid
refractive index
n20/D 1.585 (lit.)
bp
93-95 °C/0.2 mmHg (lit.)
density
1.139 g/mL at 25 °C (lit.)
작용기
sulfoxide
저장 온도
2-8°C
SMILES string
C=CS(=O)c1ccccc1
InChI
1S/C8H8OS/c1-2-10(9)8-6-4-3-5-7-8/h2-7H,1H2
InChI key
MZMJHXFYLRTLQX-UHFFFAOYSA-N
유전자 정보
human ... LOC129293(129293)
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Phenyl vinyl sulfoxide reacts with lithium enolates of ketones at -78°C in THF to yield bicyclo[n.2.0]alkan-1-ols. It also reacts with in situ generated (dialkylamino)magnesium reagent to yield symmetrical β-(dialkylamino)dithioacetals. It participates as an acetylene equivalent in Diels-Alder reactions.
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point (°F)
230.0 °F - closed cup
Flash Point (°C)
110 °C - closed cup
개인 보호 장비
Eyeshields, Gloves, multi-purpose combination respirator cartridge (US)
이미 열람한 고객
Use of phenyl vinyl sulfoxide as an acetylene equivalent in Diels-Alder cycloadditions.
Paquette LA, et al.
Journal of the American Chemical Society, 100(5), 1597-1599 (1978)
Masataka Kawakita et al.
The Journal of organic chemistry, 62(23), 8015-8017 (2001-10-24)
Vinyl sulfoxides (PhSOCR(1)=CHR(2): R(1) = H, Me, or Ph; R(2) = H or Me) were treated with (dialkylamino)magnesium reagents, generated in situ from the reaction of EtMgBr with secondary amines (R(3)R(4)NH: R(3) = Et, i-Pr, or Bn; R(4) = Me
X Ji et al.
Acta crystallographica. Section B, Structural science, 45 ( Pt 1), 93-99 (1989-02-01)
Three diastereomers of bicyclo[2.2.1]hept-5-en-2-yl phenyl sulfoxide were prepared by Diels-Alder [4 + 2] cycloadditions between phenyl vinyl sulfoxide and cyclopentadiene. The isomers were separated by column chromatography on silica gel and repeated recrystallizations gave the pure racemates of three of
Wendy A Loughlin et al.
Organic & biomolecular chemistry, 1(8), 1347-1353 (2003-08-22)
The enolates generated from cyclopentanone, cycloheptanone or cyclooctanone and LDA at -78 degrees C in THF react with (+/-)-phenyl vinyl sulfoxide under controlled conditions of temperature, reaction time, and concentration. Upon oxidation with MCPBA of the product mixtures, the novel
문서
The Diels–Alder reaction is the reaction between a conjugated diene and an alkene (dienophile) to form unsaturated six-membered rings. It is also referred to as a cycloaddition.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.