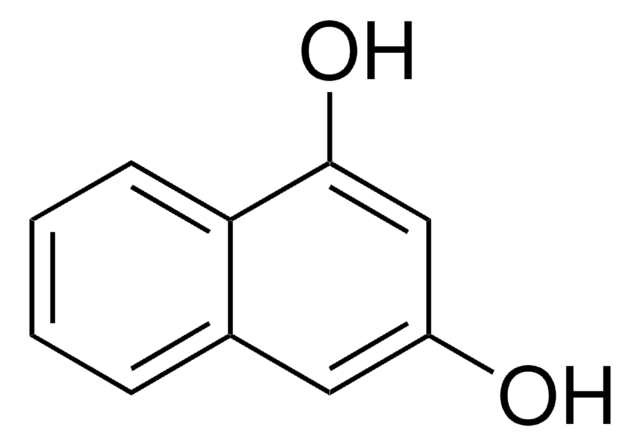
추천 제품
Quality Level
분석
98%
양식
solid
mp
223-225 °C (lit.)
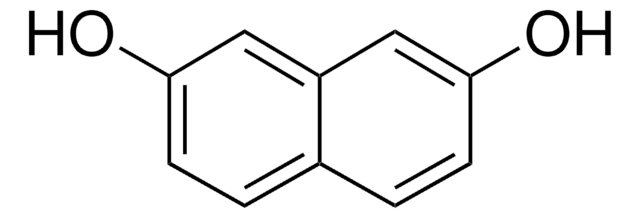
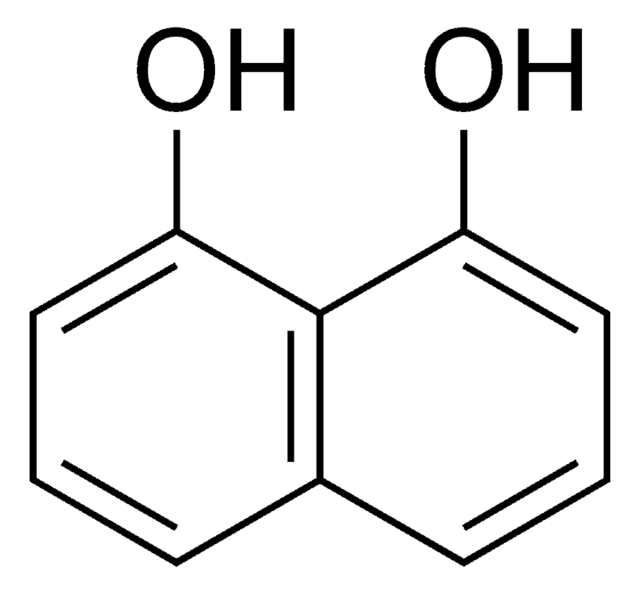
SMILES string
Oc1ccc2cc(O)ccc2c1
InChI
1S/C10H8O2/c11-9-3-1-7-5-10(12)4-2-8(7)6-9/h1-6,11-12H
InChI key
MNZMMCVIXORAQL-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
관련 카테고리
애플리케이션
2,6-Dihydroxynaphthalene was used in the synthesis of 1,5-dichloro-2,6-diethynylnaphthalenes. It was also used in the preparation of first-generation rotaxane dendrimer.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
가장 최신 버전 중 하나를 선택하세요:
시험 성적서(COA)
Lot/Batch Number
이미 열람한 고객
Shoji Shinamura et al.
The Journal of organic chemistry, 75(4), 1228-1234 (2010-01-27)
In this paper we present the synthesis, structures, characterization, and applications to field-effect transistors (FETs) of naphtho[1,2-b:5,6-b']dithiophene (NDT) and -diselenophene (NDS) derivatives. Treatment of 1,5-dichloro-2,6-diethynylnaphthalenes, easily derived from commercially available 2,6-dihydroxynaphthalene, with sodium chalcogenide afforded a straightforward access to NDTs
Soo-Young Kim et al.
Chemistry, an Asian journal, 2(6), 747-754 (2007-05-08)
By taking advantage of the fact that cucurbit[6]uril (CB[6]) forms exceptionally stable host-guest complexes with protonated amines, and that its homologue CB[8] can encapsulate a pair of electron-rich and electron-deficient guest molecules to form a stable 1:1:1 complex, we synthesized
Alberto Macone et al.
Bioorganic & medicinal chemistry, 17(16), 6003-6007 (2009-07-21)
Aromatic substrates tyrosol (p-hydroxyphenylethanol) and 2,6-dihydroxynaphthalene (2,6-DHN) were converted into chromane derivatives by means of chemoenzymatic reactions catalyzed by the aromatic prenyltransferase of bacterial origin NovQ, using dimethylallyl bromide as allylic substrate instead of the natural isoprenyl pyrophosphate substrate. Stereoselective
Mårten Jacobsson et al.
Journal of medicinal chemistry, 49(6), 1932-1938 (2006-03-17)
The antiproliferative activity of the 14 isomeric monoxylosylated dihydroxynaphthalenes has been tested in vitro toward normal HFL-1 and 3T3 A31 cells as well as transformed T24 and 3T3 SV40 cells. The antiproliferative effect toward HFL-1 cells was correlated with the
Dieter Schemeth et al.
Analytica chimica acta, 1038, 182-190 (2018-10-04)
In this study, we focus on isolation and fractionation strategies by solid phase extraction (SPE) for a broad range of environmentally related organic acids. These emerging potential contaminants are primary degradation products of spilled petrogenic compounds but little attention has
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.