348015
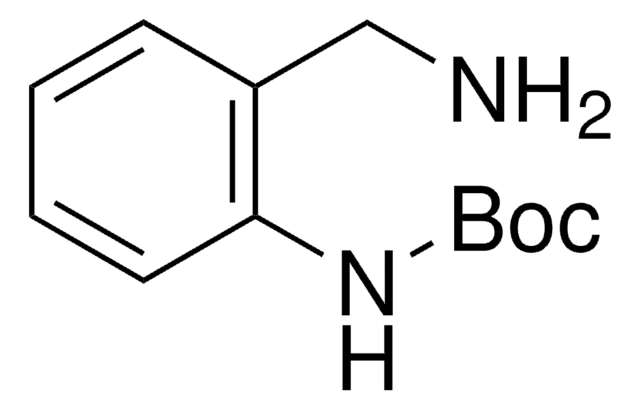
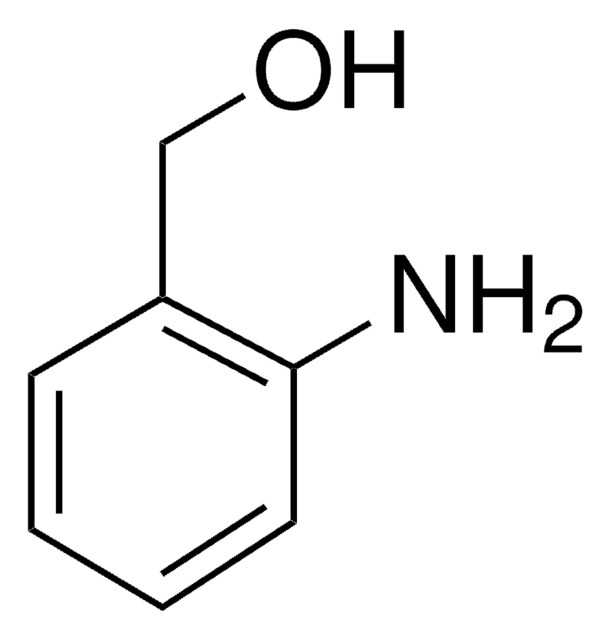
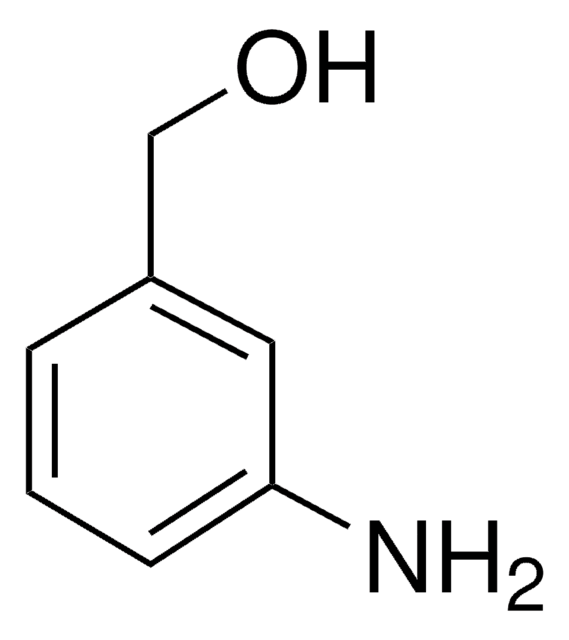
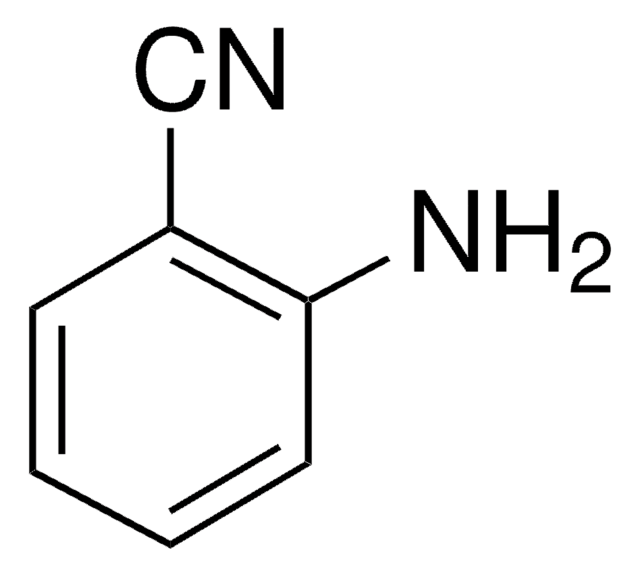
2-Aminobenzylamine
98%
동의어(들):
(2-Aminomethylphenyl)amine, 2-(Aminomethyl)aniline, 2-(Aminomethyl)benzenamine, 2-Amino-1-benzylamine, [(2-Aminophenyl)methyl]amine, o-Aminobenzylamine
로그인조직 및 계약 가격 보기
모든 사진(3)
About This Item
Linear Formula:
H2NC6H4CH2NH2
CAS Number:
Molecular Weight:
122.17
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
Quality Level
분석
98%
형태
solid
mp
58-61 °C (lit.)
작용기
amine
SMILES string
NCc1ccccc1N
InChI
1S/C7H10N2/c8-5-6-3-1-2-4-7(6)9/h1-4H,5,8-9H2
InChI key
GVOYKJPMUUJXBS-UHFFFAOYSA-N
일반 설명
2-Aminobenzylamine undergoes three-component cyclisation reactions with methyl 3,3,3-trifluoropyruvate, 2-aminobenzylamine and oxo compounds to afford regio- and stereoisomers of tetrahydropyrroloquinazolinones.
애플리케이션
2-Aminobenzylamine may be used:
- in the synthesis of 1,2,3,4-tetrahydroquinazoline oxime, via condensation reaction with 2-(naphthalen-2-yl)-2-oxoacetaldehyde oxime
- in the synthesis of alkyl 5H-1,4-benzodiazepine-3-carboxylates
- to modify the phosphate groups on phosphoserine peptides
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
이미 열람한 고객
Wen-Yun Hsueh et al.
Journal of medicinal chemistry, 64(3), 1435-1453 (2021-01-26)
In this paper, we present a copper(I)-catalyzed nitrile-addition/N-arylation ring-closure cascade for the synthesis of 5,11-dihydro-6H-indolo[3,2-c]quinolin-6-ones from 2-(2-bromophenyl)-N-(2-cyanophenyl)acetamides. Using CuBr and t-BuONa in dimethylformamide (DMF) as the optimal reaction conditions, the cascade reaction gave the target products, in high yields, with
G Sirikçi et al.
Acta chimica Slovenica, 59(4), 904-911 (2013-09-26)
A novel 1,2,3,4-tetrahydroquinazoline oxime was synthesised from a condensation reaction of 2-(naphthalen-2-yl)-2-oxoacetaldehyde oxime with 2-aminobenzylamine. Subsequently, a-imine oxime complexes of this compound that formed with Co(III) and Ni(II) metal ions were obtained. All structures were characterised by spectral methods (FT-IR
Lukas K Filak et al.
Organometallics, 30(2), 273-283 (2011-01-22)
The synthesis of new modified indolo[3,2-c]quinoline ligands L(1)-L(8) with metal-binding sites is reported. By coordination to ruthenium- and osmium-arene moieties 16 complexes of the type [(η(6)-p-cymene)M(L)Cl]Cl (1a,b-8a,b), where M is Ru(II) or Os(II) and L is L(1)-L(8), have been prepared.
Orazio A Attanasi et al.
The Journal of organic chemistry, 76(20), 8320-8328 (2011-09-13)
A novel and simple one-pot synthesis of 3-substituted 2,5,6,7-tetrahydro-1H-1,4-diazepin-2-ones from 1,2-diaza-1,3-dienes (DDs) and N-unsubstituted aliphatic 1,3-diamines is described. Here we also report a procedure to selectively obtain alkyl 5H-1,4-benzodiazepine-3-carboxylates from the DDs and 2-aminobenzylamine. Both processes occur by means of
Yu Shi et al.
Analytical chemistry, 80(19), 7614-7623 (2008-09-11)
A novel method is reported to modify the phosphate groups on phosphoserine peptides to the corresponding phosphoramidates, using 2-aminobenzylamine. Upon collision-induced dissociation, the modified peptides release the positively charged phosphoramidate that via gas-phase intramolecular elimination forms a cyclophosphoramidate (CyPAA) ion
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.