모든 사진(1)
About This Item
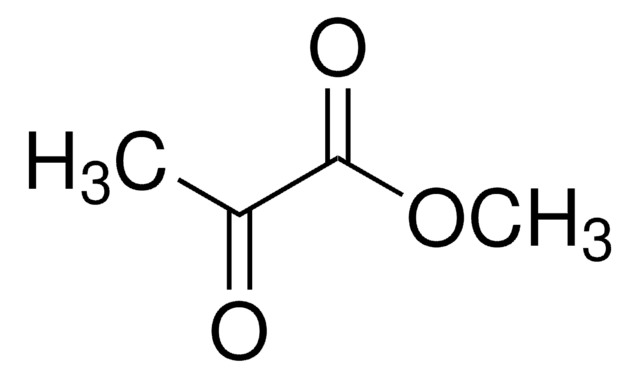
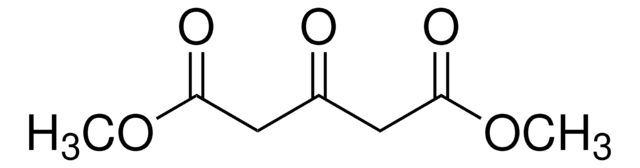
Linear Formula:
CH3O2CCH2CH2COCO2CH3
CAS Number:
Molecular Weight:
174.15
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
Quality Level
분석
96%
양식
liquid
refractive index
n20/D 1.439 (lit.)
bp
90-95 °C/0.4 mmHg (lit.)
density
1.203 g/mL at 25 °C (lit.)
작용기
ester
ketone
SMILES string
COC(=O)CCC(=O)C(=O)OC
InChI
1S/C7H10O5/c1-11-6(9)4-3-5(8)7(10)12-2/h3-4H2,1-2H3
InChI key
TXIXSLPEABAEHP-UHFFFAOYSA-N
일반 설명
Dimethyl 2-oxoglutarate is a key intermediate formed during the Krebs cycle and an important nitrogen transporter in the biological metabolic pathways. The electrochemical behavior of dimethyl-2-oxoglutarate has been investigated by cyclic voltammetry, square wave voltammetry and differential pulse voltammetry using a glassy carbon electrode.
애플리케이션
Dimethyl 2-oxoglutarate can undergo cylocondensation with dinucleophiles, such as 1,2-phenylenediamine, 2-aminophenol and 2-aminobenzenethiol to form novel heterocycles.
Dimethyl 2-oxoglutarate may be used to synthesize the conformationally constrained PNA (peptide nucleic acid) -monomer capable of binding thymine in a triplex motif. It may be used in the synthesis of 4-aryl kainic acid analogs, via highly stereoselective Michael addition reaction with nitrostyrene.
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves
이미 열람한 고객
Afzal Shah et al.
Bioelectrochemistry (Amsterdam, Netherlands), 77(2), 145-150 (2009-09-22)
The electrochemical behaviour of dimethyl-2-oxoglutarate (MOG), a key intermediate in the Krebs cycle and an important nitrogen transporter in the metabolic pathways in biological processes, was investigated by cyclic voltammetry, square wave voltammetry and differential pulse voltammetry using a glassy
Daniela Gaglio et al.
Molecular systems biology, 7, 523-523 (2011-08-19)
Oncogenes such as K-ras mediate cellular and metabolic transformation during tumorigenesis. To analyze K-Ras-dependent metabolic alterations, we employed ¹³C metabolic flux analysis (MFA), non-targeted tracer fate detection (NTFD) of ¹⁵N-labeled glutamine, and transcriptomic profiling in mouse fibroblast and human carcinoma
Organic syntheses based on 2?oxoglutaric acid. V. Syntheses of novel 2H?1, 4?benzothiazines and a 2, 5?dihydro?1, 5?benzothiazepine.
Blitzke T, et al.
Journal of Heterocyclic Chemistry, 34(2), 453-455 (1997)
An efficient synthesis of 4-aryl kainic acid analogs.
Maeda H, et al.
Tetrahedron, 55(4), 943-954 (1999)
Arun Kumar Selvam et al.
Antioxidants (Basel, Switzerland), 9(2) (2020-02-09)
Kynurenine aminotransferase 1 (KYAT1 or CCBL1) plays a major role in Se-methylselenocysteine (MSC) metabolism. It is a bi-functional enzyme that catalyzes transamination and beta-elimination activity with a single substrate. KYAT1 produces methylselenol (CH3SeH) via β-elimination activities with MSC as a
문서
Sigma article discusses tumor cell metabolic pathways, focusing on aerobic glycolysis and mitochondrial activity.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.


![1,4-Diazabicyclo[2.2.2]octane ReagentPlus®, ≥99%](/deepweb/assets/sigmaaldrich/product/structures/366/129/a6ff4175-974d-4fac-9038-b35e508ef252/640/a6ff4175-974d-4fac-9038-b35e508ef252.png)