모든 사진(1)
About This Item
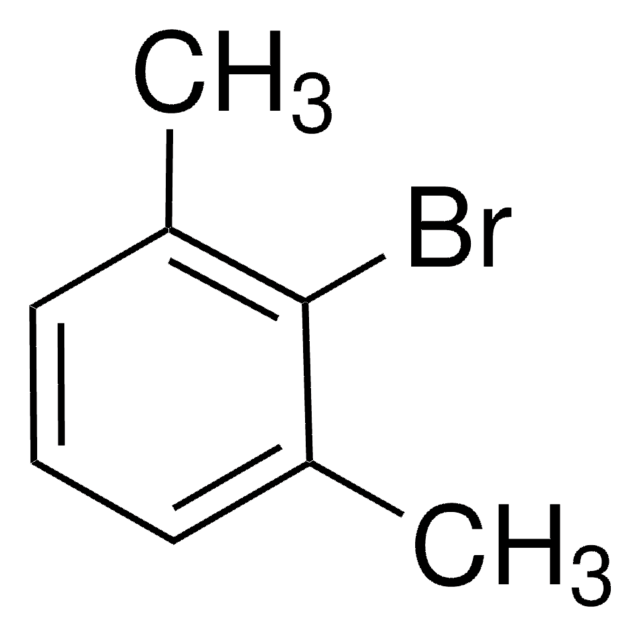
Linear Formula:
(CH3)2C6H3I
CAS Number:
Molecular Weight:
232.06
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
Quality Level
분석
99%
양식
liquid
refractive index
n20/D 1.594 (lit.)
bp
92-94 °C/3 mmHg (lit.)
density
1.608 g/mL at 25 °C (lit.)
작용기
iodo
SMILES string
Cc1cc(C)cc(I)c1
InChI
1S/C8H9I/c1-6-3-7(2)5-8(9)4-6/h3-5H,1-2H3
InChI key
ZLMKEENUYIUKKC-UHFFFAOYSA-N
일반 설명
1-Iodo-3,5-dimethylbenzene (5-Iodo-m-xylene) is an aryl halide. It can be obtained from 5-bromo-m-xylene, via copper-catalyzed halogen exchange reaction, in the presence of NaI or KI in n-BuOH or DMF (solvents). It undergoes reaction with phenol in the presence of CuFe2O4 nano powder as a recyclable catalyst to afford 1,3-dimethyl-5-phenoxybenzene.
애플리케이션
1-Iodo-3,5-dimethylbenzene (5-iodo-m-xylene) is suitable for use in the synthesis of N-(3,5-xylyl)-N-ethylaniline, an arylamine.
It may be used in the following studies:
It may be used in the following studies:
- α-Arylation of ketones.
- Copper-catalyzed N-arylation of imidazoles.
- Cyanation of 5-iodo-m-xylene to form 3,5-dimethylbenzonitrile.
- Synthesis of 1,3-Dimethyl-5-phenoxybenzene by nano-CuFe2O4 catalyzed C-O cross-coupling with phenol.
- CuBr-catalyzed amination of 1-iodo-3,5-dimethylbenzene to form N-Allyl-3,5-dimethylbenzenamine.
- Copper-catalyzed C-S bond-formation between 5-iodo-m-xylene and thiophenol.
- As a starting material in the synthesis of biphenyl-3,3′,5,5′-tetracarboxylic acid.
- Radical bromination of 5-iodo-m-xylene by N-bromosuccinimide to form 1,3-bis(bromomethyl)-5-iodobenzene.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point (°F)
235.4 °F - closed cup
Flash Point (°C)
113 °C - closed cup
개인 보호 장비
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
이미 열람한 고객
Biphenyl-3, 3', 5, 5'-tetracarboxylic acid.
Coles SJ, et al.
Acta Crystallographica Section E, Structure Reports Online, 58(6), 626-628 (2002)
On the synthesis of heterocyclic dendrons.
Diez-Barra E, et al.
ARKIVOC (Gainesville, FL, United States), 2002(5), 17-25 (2002)
Jacopo Zanon et al.
Journal of the American Chemical Society, 125(10), 2890-2891 (2003-03-06)
An efficient copper-catalyzed domino halogen exchange-cyanation procedure for aryl bromides was developed utilizing 10 mol % CuI, 20 mol % KI, 1.0 equiv of the inexpensive N,N'-dimethylethylenediamine as ligand, and 1.2 equiv of NaCN in toluene at 110 degrees C.
Recyclable and reusable nano-CuFe2O4 catalyzed CO cross-coupling.
Avudoddi V, et al.
European Journal of Chemistry, 3(3), 298-304 (2012)
M H Ali et al.
The Journal of organic chemistry, 66(8), 2560-2565 (2001-04-17)
Aryl iodides are coupled with amines to give the corresponding arylamines in high yield in the presence of palladium, a suitable ligand, and NaOt-Bu. Functionalized aryl iodides give good yields of the corresponding arylamines when Cs(2)CO(3) is substituted as the
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.