57410
Indole-3-propionic acid
≥97.0% (T)
동의어(들):
NSC 3252, NSC 47831, 3-(3-Indolyl)propanoic acid, 3-(3-Indolyl)propionic acid, IPA
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
실험식(Hill 표기법):
C11H11NO2
CAS Number:
Molecular Weight:
189.21
Beilstein:
147733
EC Number:
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
Quality Level
분석
≥97.0% (T)
양식
solid
작용기
carboxylic acid
SMILES string
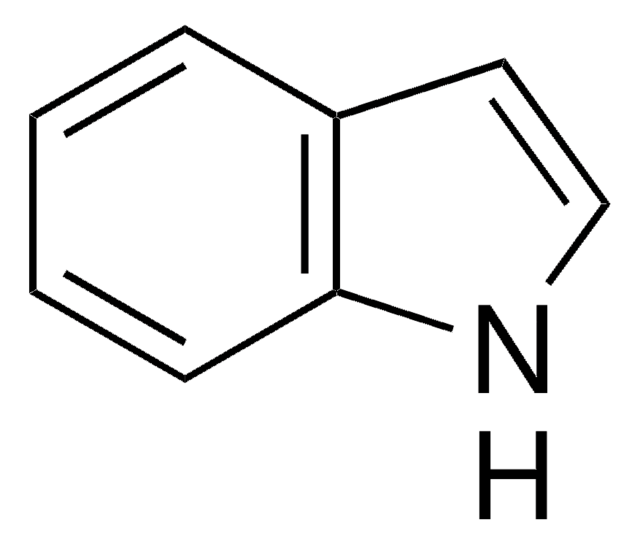
OC(=O)CCc1c[nH]c2ccccc12
InChI
1S/C11H11NO2/c13-11(14)6-5-8-7-12-10-4-2-1-3-9(8)10/h1-4,7,12H,5-6H2,(H,13,14)
InChI key
GOLXRNDWAUTYKT-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Indole-3-propionic acid can be obtained from tryptophan by deamination reaction.
애플리케이션
Reactant for preparation of:
- Fluorescent analogues of strigolactones
- Anti-tumor agents
- Melanocortin receptors ligands
- Immunosuppressive agents
- Iinhibitors of hepatitis C virus
- Histamine H4 receptor agonists
- NR2B/NMDA receptor antagonists
- CB1 antagonist for the treatment of obesity
- Antibacterial agents
- Inhibitor of TGF-β receptor binding
생화학적/생리학적 작용
Studied as an adjunct to improve perfusion after liver transplant.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
Design and parallel synthesis of new bicyclic small molecules for targeting the melanocortin receptors.
Byoung J Min et al.
Advances in experimental medicine and biology, 611, 187-188 (2009-04-30)
Prandi, C.; et al.
European Journal of Organic Chemistry, 3781-3781 (2011)
Rosaria Gitto et al.
Bioorganic & medicinal chemistry, 17(4), 1640-1647 (2009-01-23)
A combined ligand-based and structure-based approach has previously allowed us to identify NR2B/NMDA receptor antagonists containing indole scaffold. In order to further explore the main structure activity relationships of this class of derivatives we herein report the design, synthesis and
Subra, G.; et al.
QSAR & Combinatorial Science, 26, 496-496 (2007)
Francis Giraud et al.
Bioorganic & medicinal chemistry letters, 20(17), 5203-5206 (2010-07-27)
N-aryl-3-(indol-3-yl)propanamides were synthesized and their immunosuppressive activities were evaluated. This study highlighted the promising potency of 3-[1-(4-chlorobenzyl)-1H-indol-3-yl]-N-(4-nitrophenyl)propanamide 15 which exhibited a significant inhibitory activity on murine splenocytes proliferation assay in vitro and on mice delayed-type hypersensitivity (DTH) assay in vivo.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.