추천 제품
Quality Level
분석
98%
형태
powder
mp
274-276 °C (dec.) (lit.)
작용기
iodo
저장 온도
2-8°C
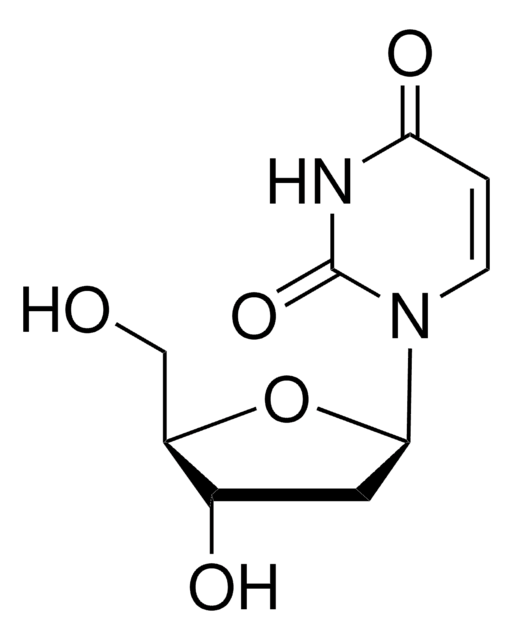
SMILES string
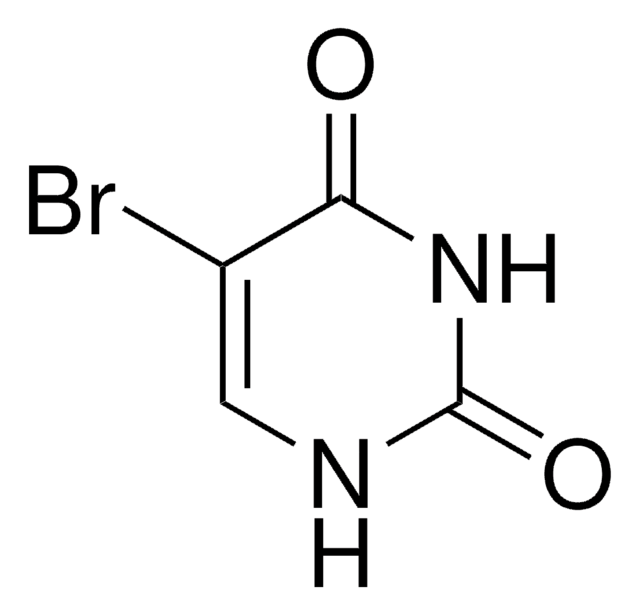
IC1=CNC(=O)NC1=O
InChI
1S/C4H3IN2O2/c5-2-1-6-4(9)7-3(2)8/h1H,(H2,6,7,8,9)
InChI key
KSNXJLQDQOIRIP-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
13 - Non Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type P2 (EN 143) respirator cartridges
Doreen Dobritzsch et al.
The Journal of biological chemistry, 277(15), 13155-13166 (2002-01-18)
Dihydroprymidine dehydrogenase catalyzes the first and rate-limiting step in pyrimidine degradation by converting pyrimidines to the corresponding 5,6- dihydro compounds. The three-dimensional structures of a binary complex with the inhibitor 5-iodouracil and two ternary complexes with NADPH and the inhibitors
D L Wong et al.
Nucleic acids research, 26(2), 645-649 (1998-02-28)
We describe a novel strategy combining photocrosslinking and HPLC-based electrospray ionization mass spectrometry to identify UV crosslinked DNA-protein complexes. Eco RI DNA methyltransferase modifies the second adenine within the recognition sequence GAATTC. Substitution of 5-iodouracil for the thymine adjacent to
Yan Xu et al.
Journal of the American Chemical Society, 126(20), 6274-6279 (2004-05-20)
To explore the structure-dependent hydrogen abstraction in antiparallel and parallel G-quartet DNA structures, the photochemical reactivity of 5-iodouracil ((I)U)-containing human telomeric DNA 22-mers was investigated under the 302 nm UV irradiation conditions. We discovered that only antiparallel ODN 4, in
Supaluk Prachayasittikul et al.
Molecules (Basel, Switzerland), 14(8), 2768-2779 (2009-08-25)
This study reports the synthesis of some substituted 5-iodouracils and their bioactivities. Alkylation of 5-iodouracils gave predominately N1-substituted-(R)-5-iodouracil compounds 7a-d (R = n-C(4)H(9), s-C(4)H(9), CH(2)C(6)H(11), CH(2)C(6)H(5)) together with N1,N3-disubstituted (R) analogs 8a-b (R = n-C(4)H(9), CH(2)C(6)H(11)). Their antimicrobial activity was
M C Willis et al.
Science (New York, N.Y.), 262(5137), 1255-1257 (1993-11-19)
5-Iodouracil-substituted RNA and DNA were crosslinked regiospecifically to associated proteins in yields of 70 to 94% of bound nucleic acid. Irradiation of the iodouracil chromophore with monochromatic, long-wavelength ultraviolet radiation (325 nanometers) eliminates excitation of other nucleic acid and protein
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.