추천 제품
분석
99%
형태
powder
mp
117-119 °C (lit.)
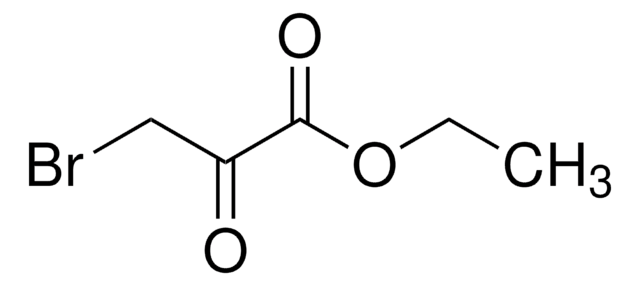
SMILES string
BrCC(=O)C(=O)CBr
InChI
1S/C4H4Br2O2/c5-1-3(7)4(8)2-6/h1-2H2
InChI key
RZMOICJDRADLCT-UHFFFAOYSA-N
애플리케이션
- Catalyst-Free Multicomponent Cyclopolymerizations: A novel approach utilizing 1,4-Dibromo-2,3-butanedione in catalyst-free multicomponent cyclopolymerizations to synthesize functional polyiminofurans containing bromomethyl groups was developed. This method enhances the utility of 1,4-Dibromo-2,3-butanedione in producing advanced materials with potential applications in chemical and pharmaceutical industries (Zhu et al., 2021).
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
S H Vollmer et al.
Protein science : a publication of the Protein Society, 1(5), 678-687 (1992-05-01)
The bifunctional reagent 1,4-dibromobutanedione (DBBD) reacts covalently with pyruvate kinase from rabbit muscle to cause inactivation of the enzyme at a rate that is linearly dependent on the reagent concentration, giving a second order rate constant of 444 min-1 M-1.
Chui-Shan Tsang et al.
Chemical communications (Cambridge, England), (15)(15), 1999-2001 (2009-04-01)
Reaction of a pinene-based pyridylthioamide with 1,4-dibromo-2,3-butanedione in refluxing methanol yielded a new chiral pyridylthiazole ligand L which forms a dinuclear double-stranded helicate with Cu(i) ions; this helicate has opposite helical chirality when compared with its quaterpyridine analogue.
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.