M13203
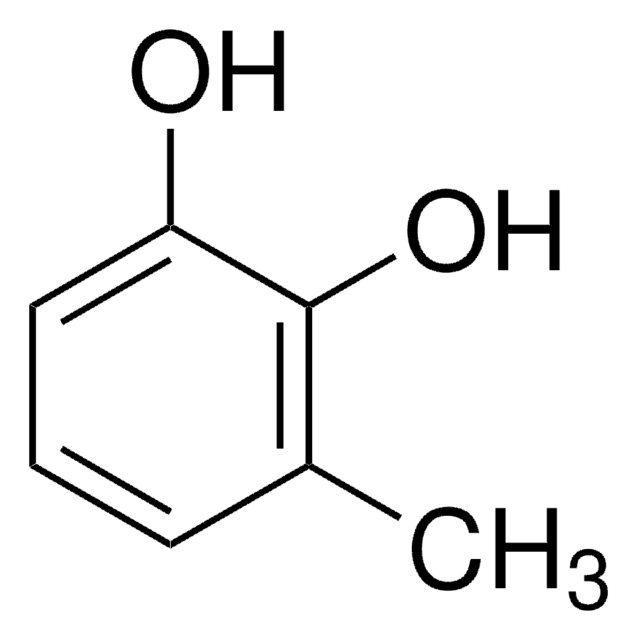
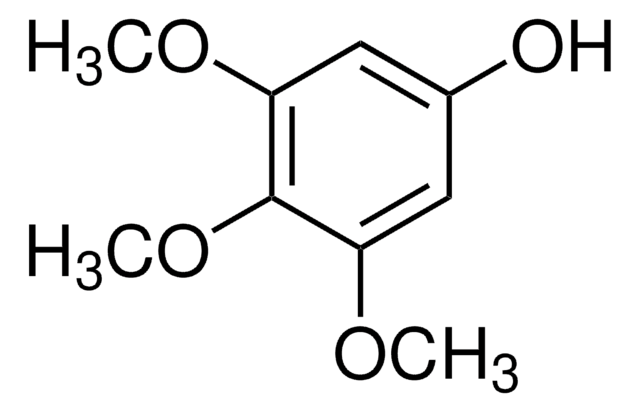
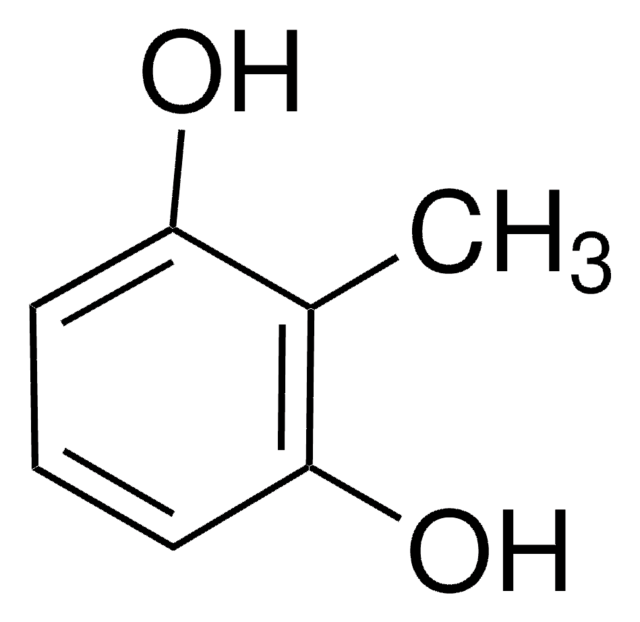
3-Methoxycatechol
99%
동의어(들):
1,2-Dihydroxy-3-methoxybenzene, 3-Methoxypyrocatechol, Pyrogallol monomethyl ether
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
Linear Formula:
CH3OC6H3(OH)2
CAS Number:
Molecular Weight:
140.14
Beilstein:
1909165
EC Number:
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
Quality Level
분석
99%
bp
146-147 °C/15 mmHg (lit.)
mp
38-43 °C (lit.)
SMILES string
COc1cccc(O)c1O
InChI
1S/C7H8O3/c1-10-6-4-2-3-5(8)7(6)9/h2-4,8-9H,1H3
InChI key
LPYUENQFPVNPHY-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
관련 카테고리
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
이미 열람한 고객
M Kawabe et al.
Japanese journal of cancer research : Gann, 85(1), 17-25 (1994-01-01)
The effects of combined treatment with NaNO2 and phenolic compounds on N-methyl-N'-nitro-N-nitrosoguanidine (MNNG) stomach carcinogenesis were investigated in F344 rats. In the first experiment, groups of 15-20 male rats were treated with an intragastric dose of 150 mg/kg body weight
M Hirose et al.
Japanese journal of cancer research : Gann, 81(9), 857-861 (1990-09-01)
The effects of sodium nitrite (NaNO2) and catechol or 3-methoxycatechol in combination were examined in male F344 rats. Animals were treated with 0.3% NaNO2 in the drinking water and 0.8% catechol or 2% 3-methoxycatechol in powdered diet for 24 weeks.
M Hirose et al.
Cancer research, 53(1), 32-37 (1993-01-01)
Effects of simultaneous treatment with NaNO2 and butylated hydroxyanisole, catechol, or 3-methoxycatechol were examined in a rat multiorgan carcinogenesis model. Groups of 15 animals were given a single i.p. injection of 100 mg/kg of body weight diethylnitrosamine, 4 i.p. injections
Huayun Deng et al.
Pharmaceuticals (Basel, Switzerland), 6(4), 500-509 (2013-11-28)
Nearly 1% of all clinically used drugs are catecholics, a family of catechol-containing compounds. Using label-free dynamic mass redistribution and Tango β-arrestin translocation assays, we show that several catecholics, including benserazide, catechol, 3-methoxycatechol, pyrogallol, (+)-taxifolin and fenoldopam, display agonistic activity
Keith M Anderson et al.
The Journal of organic chemistry, 72(26), 9875-9880 (2007-11-30)
A novel universal support for deoxyribo- and ribonucleic acid synthesis has been developed. The support, constructed from 1,4-dimethoxycatechol, represents an improvement over existing universal supports because of its ability to cleave and deprotect under mild conditions in standard reagents. Because
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.