T13005
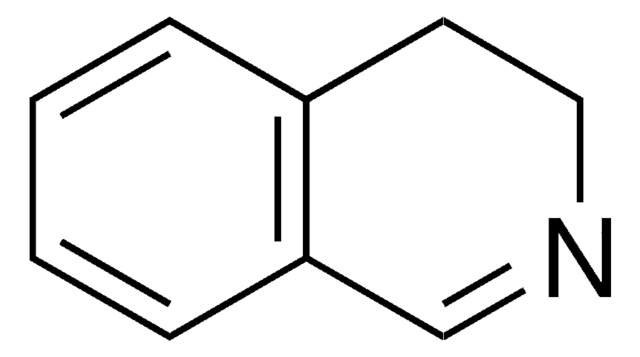
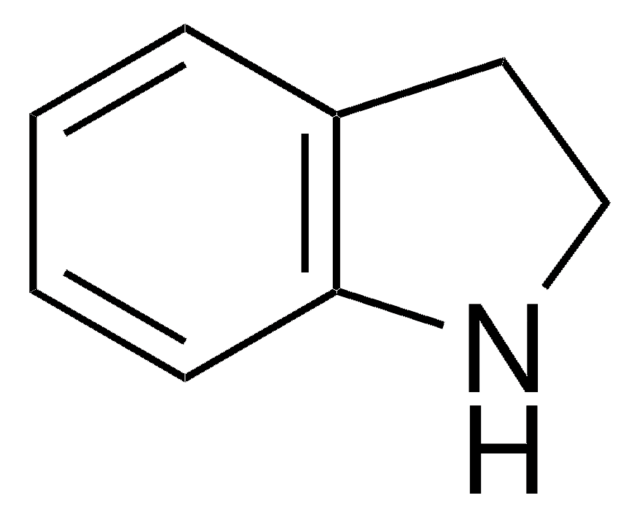
1,2,3,4-Tetrahydroisoquinoline
95%
동의어(들):
1,2,3,4-Tetrahydro-2-azanaphthalene, 1,2,3,4-Tetrahydro-2-isoquinoline, 1,2,3,4-Tetrahydroisoquinline, 1,2,3,4-Tetrahydroisoquinoline, 3,4-Dihydro-1H-isoquinoline
About This Item
추천 제품
Quality Level
분석
95%
refractive index
n20/D 1.568 (lit.)
bp
232-233 °C (lit.)
mp
−30 °C (lit.)
density
1.064 g/mL at 25 °C (lit.)
SMILES string
C1Cc2ccccc2CN1
InChI
1S/C9H11N/c1-2-4-9-7-10-6-5-8(9)3-1/h1-4,10H,5-7H2
InChI key
UWYZHKAOTLEWKK-UHFFFAOYSA-N
유전자 정보
human ... PNMT(5409)
rat ... Adra2a(25083) , Htr1a(24473)
유사한 제품을 찾으십니까? 방문 제품 비교 안내
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 2 Dermal - Acute Tox. 3 Oral - Acute Tox. 4 Inhalation - Aquatic Chronic 3 - Eye Dam. 1 - Skin Corr. 1B - STOT SE 2
Storage Class Code
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 3
Flash Point (°F)
210.2 °F - closed cup
Flash Point (°C)
99 °C - closed cup
개인 보호 장비
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
이미 열람한 고객
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.