W421501
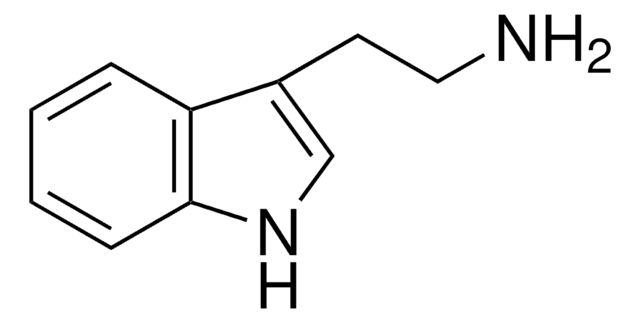
Tyramine
98%, FG
동의어(들):
2-(4-Hydroxyphenyl)ethylamine, 4-(2-Aminoethyl)phenol, 4-Hydroxyphenethylamine
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
Linear Formula:
HOC6H4CH2CH2NH2
CAS Number:
Molecular Weight:
137.18
FEMA Number:
4215
Beilstein:
1099914
EC Number:
MDL number:
UNSPSC 코드:
12164502
PubChem Substance ID:
플래비스(Flavis) 번호:
11.007
NACRES:
NA.21
감각 수용성의:
meaty; phenolic
Grade:
FG
생물학적 소스:
synthetic
식품 알레르기항원:
no known allergens
추천 제품
생물학적 소스
synthetic
Quality Level
Grade
FG
규정 준수
EU Regulation 1334/2008 & 872/2012
FDA 21 CFR 110
분석
98%
bp
175-181 °C/8 mmHg (lit.)
mp
160-162 °C (lit.)
응용 분야
flavors and fragrances
문건
see Safety & Documentation for available documents
식품 알레르기항원
no known allergens
감각 수용성의
meaty; phenolic
SMILES string
NCCc1ccc(O)cc1
InChI
1S/C8H11NO/c9-6-5-7-1-3-8(10)4-2-7/h1-4,10H,5-6,9H2
InChI key
DZGWFCGJZKJUFP-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
관련 카테고리
생화학적/생리학적 작용
Can enter catecholaminergic terminals and be released as a false transmitter.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
이미 열람한 고객
Stephan A Chalon et al.
Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology, 28(9), 1685-1693 (2003-06-05)
Evidence suggests that compounds that increase the synaptic availability of more than one neurotransmitter have greater efficacy in the treatment of depression than single-acting drugs. Preclinical studies indicate that duloxetine acts to inhibit serotonin (5-HT) and norepinephrine (NE) transporters. The
Giris Jacob et al.
Circulation, 107(19), 2475-2479 (2003-04-23)
Despite the widespread use of tyramine as a pharmacological tool to assess the effects of norepinephrine release from sympathetic nerve terminals, its vascular effects are not adequately characterized. In particular, previous results indicate that intravenous tyramine produces little if any
D R Varma et al.
The Journal of pharmacology and experimental therapeutics, 265(3), 1096-1104 (1993-06-01)
At concentrations higher than that required to produce maximal vasoconstriction, tyramine caused concentration-dependent relaxation of rat aortic strips contracted maximally by tyramine, norepinephrine, phenylephrine, 5-hydroxytryptamine, prostaglandin F2 alpha, endothelin, angiotensin II and potassium; isoproterenol did not relax potassium-contracted strips. The
Thomas Roeder
Annual review of entomology, 50, 447-477 (2004-09-10)
Octopamine (OA) and tyramine (TA) are the invertebrate counterparts of the vertebrate adrenergic transmitters. They are decarboxylation products of the amino acid tyrosine, with TA as the biological precursor of OA. Nevertheless, both compounds are independent neurotransmitters that act through
Moussa B H Youdim et al.
Neurotoxicology, 25(1-2), 243-250 (2003-12-31)
The major side effect with the use of first generation of non selective monoamine oxidase (MAO) inhibitors as neuropsychiatric drugs was what became known as the "cheese reaction". Namely, potentiation of sympathomimetic activity of ingested tyramine present in cheese and
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.