PHR1148
4-Aminophenol (Acetaminophen Related Compound K) (Paracetamol Impurity K)
Pharmaceutical Secondary Standard; Certified Reference Material
동의어(들):
4-Aminophenol, 4-Hydroxyaniline
About This Item
추천 제품
Grade
certified reference material
pharmaceutical secondary standard
Quality Level
Agency
traceable to Ph. Eur. Y0001955
traceable to USP 1021204
API family
mesalazine, paracetamol, acetaminophen
CofA
current certificate can be downloaded
기술
HPLC: suitable
gas chromatography (GC): suitable
mp
185-189 °C (lit.)
응용 분야
pharmaceutical (small molecule)
형식
neat
저장 온도
2-30°C
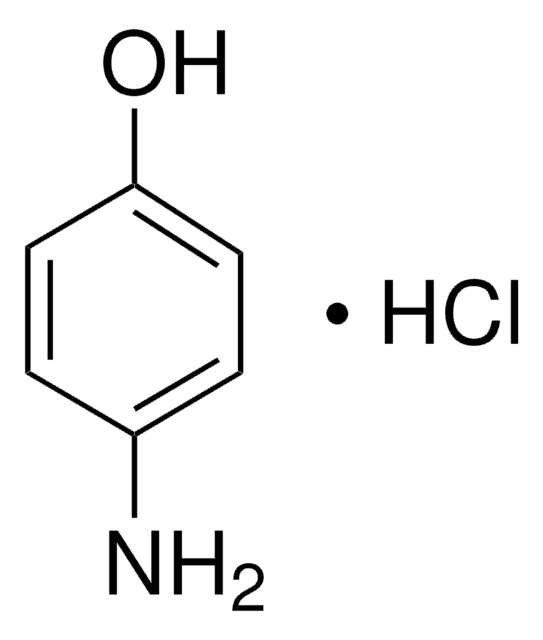
SMILES string
Nc1ccc(O)cc1
InChI
1S/C6H7NO/c7-5-1-3-6(8)4-2-5/h1-4,8H,7H2
InChI key
PLIKAWJENQZMHA-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
It is a primary degradation product of the widely used antipyretic and analgesic drug acetaminophen, also known as paracetamol, formed on its hydrolysis.
애플리케이션
- Separation and estimation of acetaminophen and its process impurities in commercial acetaminophen tablets using high-performance liquid chromatography (HPLC)
- Simultaneous electrochemical determination of acetaminophen and its impurity K using a reduced graphene oxide-titanium nitride (RGO-TiN) nanohybrid
- Development and validation of HPLC-based stability indicating method for the determination of acetaminophen, chlorpheniramine maleate, and their possible degradation products in an over-the-counter syrup formulation
- Determination of acetaminophen, pamabrom, and their impurities using high-performance thin layer chromatography (HPTLC) and reversed phase-high performance liquid chromatography (RP-HPLC), according to the ICH guidelines, in their combined dosage tablets
- Combined analysis of acetaminophen, chlorzoxazone, and their major degradation impurities using thin-layer chromatographic (TLC)-densitometric method in their combined dosage capsules
분석 메모
각주
추천 제품
관련 제품
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Muta. 2 - Skin Sens. 1 - STOT RE 2
표적 기관
Kidney
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
이미 열람한 고객
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.