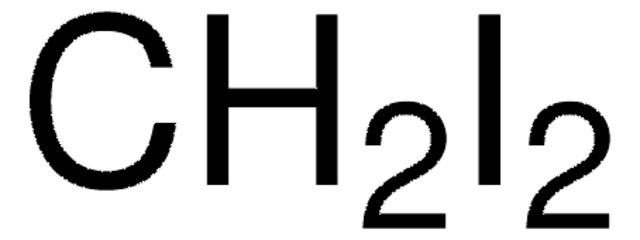모든 사진(1)
About This Item
실험식(Hill 표기법):
C37H37ClN4O10
Molecular Weight:
733.16
MDL number:
UNSPSC 코드:
12352111
NACRES:
NA.32
추천 제품
제품 라인
BioReagent
Quality Level
분석
>90.0% (HPLC)
형태
powder
제조업체/상표
ATTO-TEC GmbH
투과
254 nm
565 nm
형광
λex 563 nm; λem 592 nm in 0.1 M phosphate pH 7.0
λ
(ethanol with 0.1% trifluoroacetic acid)
UV 흡수
λ: 560-566 nm Amax
적합성
corresponds for coupling to thiols
suitable for fluorescence
저장 온도
−20°C
애플리케이션
Atto 565 is a new label with high molecular absorption (120.000) and quantum yield (0.9) as well as sufficient stoke′s shift (excitation maximum 563 nm, emission maximum 592 nm). Due to an insignificant triplet formation rate it is well suited for single molecule detection applications. Maleimides are well suited for coupling to thiol groups. This is similar to iodacetamides, but maleimides do react more thiol selective. They do not show significant reaction with histidine or methionine. Hydrolysis of maleimides to a mixture of isomeric nonreactive maleamic acids can compete significantly with thiol modification, particularly above pH 8. Maleimides may be used for labeling of amines, which usually requires a higher pH than reaction of maleimides with thiols.
법적 정보
This product is for Research use only. In case of intended commercialization, please contact the IP-holder (ATTO-TEC GmbH, Germany) for licensing.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
이미 열람한 고객
Shadi Ferdosi et al.
Journal of proteome research, 17(1), 543-558 (2017-11-14)
Glycans represent a promising but only marginally accessed source of cancer markers. We previously reported the development of a molecularly bottom-up approach to plasma and serum (P/S) glycomics based on glycan linkage analysis that captures features such as α2-6 sialylation
Lukasz Krzemiński et al.
The journal of physical chemistry. B, 115(43), 12607-12614 (2011-09-24)
Recently, studies have been reported in which fluorescently labeled redox proteins have been studied with a combination of spectroscopy and electrochemistry. In order to understand the effect of the dye on the protein-electrode interaction, voltammetry and surface analysis have been
Thiol reactive probes and chemosensors.
Peng H, Chen W, Cheng Y, et al.
Sensors, 12, 15907-15946 (2012)
Suman Lata et al.
Journal of the American Chemical Society, 128(7), 2365-2372 (2006-02-16)
Labeling of proteins with fluorescent dyes offers powerful means for monitoring protein interactions in vitro and in live cells. Only a few techniques for noncovalent fluorescence labeling with well-defined localization of the attached dye are currently available. Here, we present
Limin Ma et al.
The Analyst, 137(21), 5046-5050 (2012-09-13)
4-Nitro-1,8-naphthalic anhydride (NNA) was used to distinguish cysteine from homocysteine and other potentially interfering thiols through a novel sequential substitution mechanism. The discrimination involves a blue-fluorescent thioether formation via nucleophilic aromatic substitution of the nitro group by thiol, followed by
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.