모든 사진(1)
About This Item
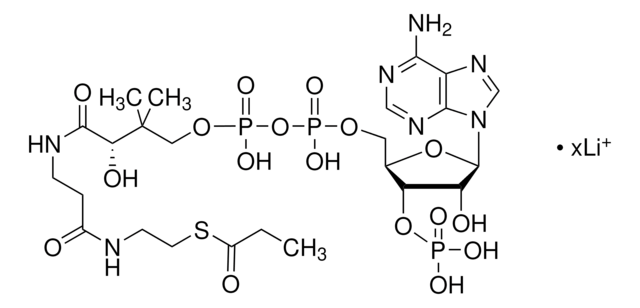
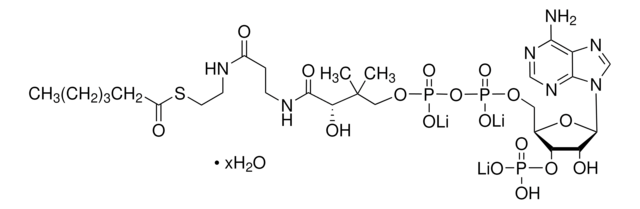
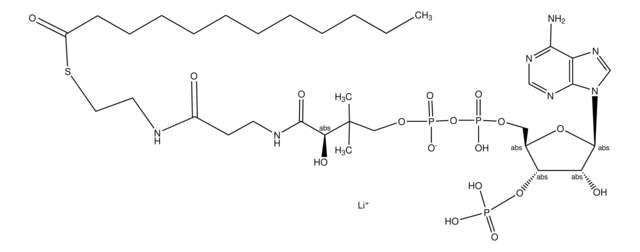
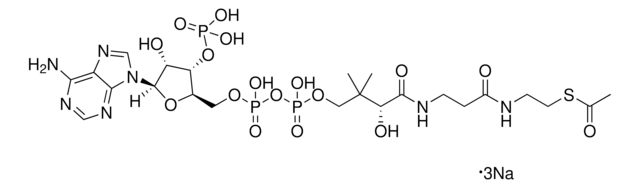
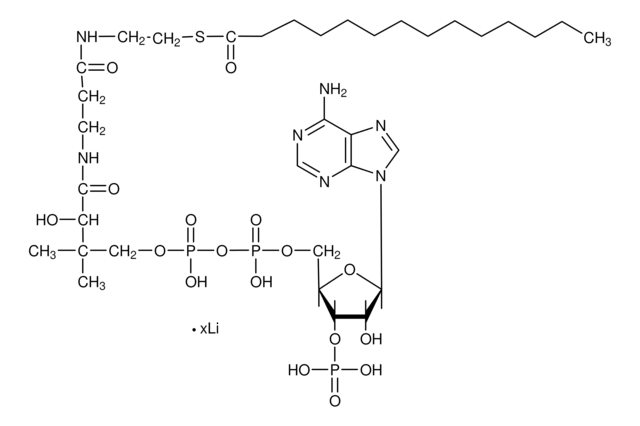
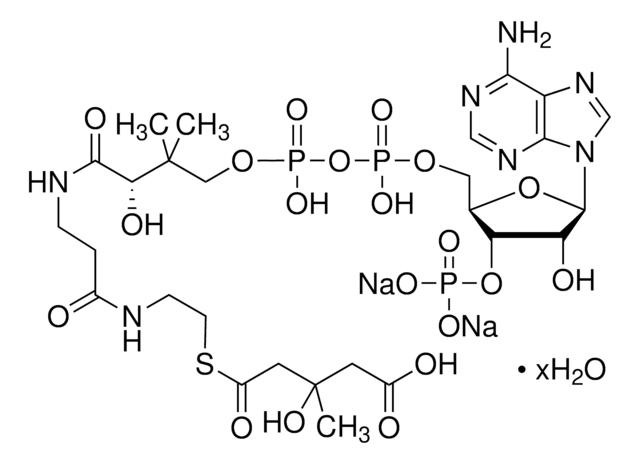
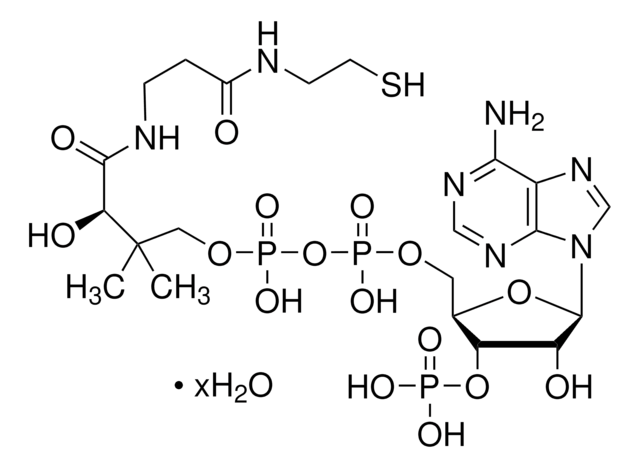
실험식(Hill 표기법):
C28H40N7O17P3S
CAS Number:
Molecular Weight:
871.64
MDL number:
UNSPSC 코드:
41106305
PubChem Substance ID:
NACRES:
NA.51
추천 제품
Quality Level
분석
≥90%
양식
solid
저장 온도
−20°C
SMILES string
[Li].CC(C)(COP(O)(=O)OP(O)(=O)OCC1OC(C(O)C1OP(O)(O)=O)n2cnc3c(N)ncnc23)C(O)C(=O)NCCC(=O)NCCSC(=O)c4ccccc4
InChI
1S/C28H40N7O17P3S.Li.H/c1-28(2,22(38)25(39)31-9-8-18(36)30-10-11-56-27(40)16-6-4-3-5-7-16)13-49-55(46,47)52-54(44,45)48-12-17-21(51-53(41,42)43)20(37)26(50-17)35-15-34-19-23(29)32-14-33-24(19)35;;/h3-7,14-15,17,20-22,26,37-38H,8-13H2,1-2H3,(H,30,36)(H,31,39)(H,44,45)(H,46,47)(H2,29,32,33)(H2,41,42,43);;
InChI key
HBWPXONQNHVILU-UHFFFAOYSA-N
일반 설명
Benzoyl coenzyme A (Benzoyl CoA) is an intermediate in the CoA-dependent epoxide pathway, synthesized from benzoate in the presence of the enzyme benzoate-CoA ligase. It is further converted to 2,3-epoxybenzoyl-CoA and this step is catalyzed by benzoyl-CoA reductase (BoxA) and benzoyl-CoA oxygenase(BoxB).
생화학적/생리학적 작용
Benzoyl coenzyme A (Benzoyl CoA) is a substrate of benzoyl-CoA reductase (box A) and a starter substrate for type-III polyketide synthase. It also acts as a substrate for plant enzymes biphenyl synthase (BIS) and benzophenone synthase (BPS). Benzoyl CoA is an effector of gentisate pathway of the bacteria, Comamonas testosteroni. It may also be used as a substrate for the characterization of specific alcohol acyltransferases and N-acyltransferase assays.
Benzoyl coenzyme A (Benzoyl CoA) may be used in studies of benzoate metabolism. Benzoyl CoA is a component of benzoyl-coenzyme A (CoA) oxidizing epoxidase enzyme system BoxAB. Benzoyl CoA may also be used as a substrate for characterization of specific alcohol acyltransferases. Benzoyl-CoA may be used as a starter substrate for synthesis of biphenyl and dibenzofuran phytoalexins (polyketide derivatives).
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
가장 최신 버전 중 하나를 선택하세요:
시험 성적서(COA)
Lot/Batch Number
이미 열람한 고객
Cornelia Chizzali et al.
Beilstein journal of organic chemistry, 8, 613-620 (2012-05-09)
Biphenyls and dibenzofurans are the phytoalexins of the Pyrinae, a subtribe of the plant family Rosaceae. The Pyrinae correspond to the long-recognized Maloideae. Economically valuable species of the Pyrinae are apples and pears. Biphenyls and dibenzofurans are formed de novo
A versatile biosynthetic approach to amide bond formation
Philpott HK, et al.
Green Chemistry, 20(15), 3426-3431 (2018)
Catrin S Günther et al.
Phytochemistry, 72(8), 700-710 (2011-04-01)
Volatile esters are key compounds of kiwifruit flavour and are formed by alcohol acyltransferases that belong to the BAHD acyltransferase superfamily. Quantitative RT-PCR was used to screen kiwifruit-derived expressed sequence tags with proposed acyltransferase function in order to select ripening-specific
Liv J Rather et al.
The Journal of biological chemistry, 286(33), 29241-29248 (2011-06-03)
The coenzyme A (CoA)-dependent aerobic benzoate metabolic pathway uses an unprecedented chemical strategy to overcome the high aromatic resonance energy by forming the non-aromatic 2,3-epoxybenzoyl-CoA. The crucial dearomatizing reaction is catalyzed by three enzymes, BoxABC, where BoxA is an NADPH-dependent
Liv J Rather et al.
Biochimica et biophysica acta, 1814(12), 1609-1615 (2011-06-16)
BoxA is the reductase component of the benzoyl-coenzyme A (CoA) oxidizing epoxidase enzyme system BoxAB. The enzyme catalyzes the key step of an hitherto unknown aerobic, CoA-dependent pathway of benzoate metabolism, which is the epoxidation of benzoyl-CoA to the non-aromatic
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.