129488
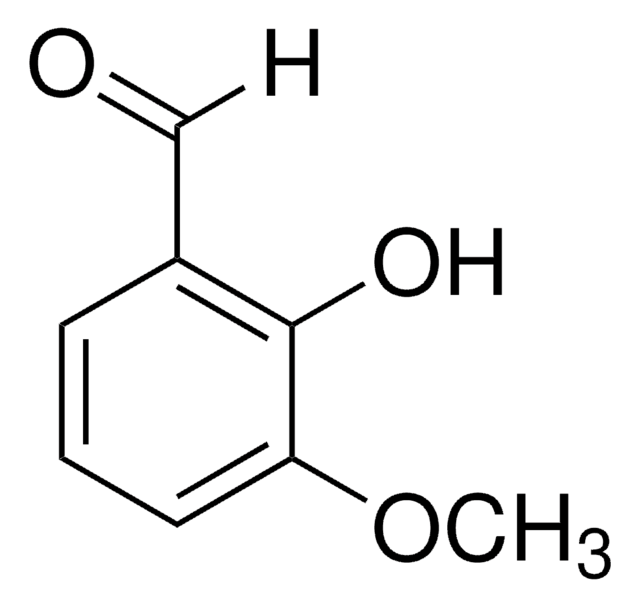
5-Iodovanillin
97%
Synonym(s):
4-Hydroxy-3-iodo-5-methoxybenzaldehyde
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
IC6H2(OCH3)(OH)CHO
CAS Number:
Molecular Weight:
278.04
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
97%
form
powder
mp
183-185 °C (lit.)
functional group
aldehyde
iodo
SMILES string
COc1cc(C=O)cc(I)c1O
InChI
1S/C8H7IO3/c1-12-7-3-5(4-10)2-6(9)8(7)11/h2-4,11H,1H3
InChI key
FBBCSYADXYILEH-UHFFFAOYSA-N
Application
5-Lodovanillin was used in the characterization of 5-Iodoacetovanillone. 5-Iodovanillin has also been used in the synthesis of platelet activating factor antagonist L-659,989.
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
An improved synthesis of acetosyringone.
Crawford LW, et al.
Canadian Journal of Chemistry, 34(11), 1562-1566 (1956)
Thompson AS, et al.
Tetrahedron Letters, 31(48), 6953-6956 (1990)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service