179825
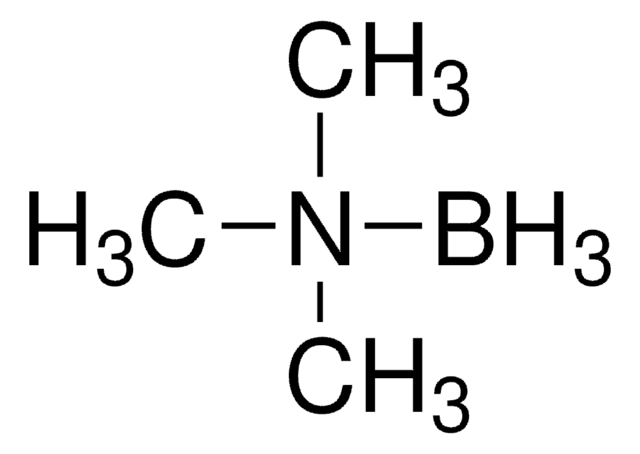
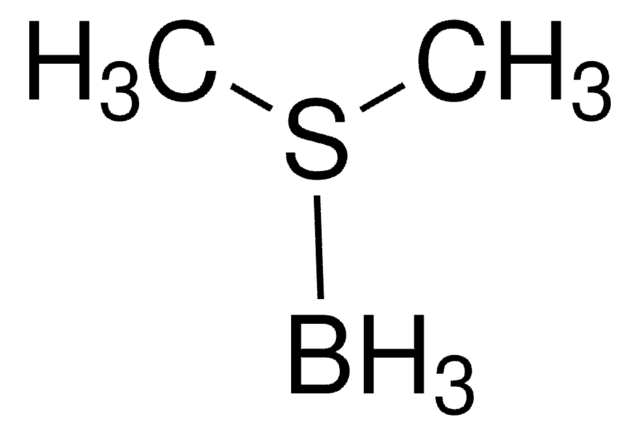
Borane dimethyl sulfide complex
Synonym(s):
(Dimethyl sulfide)trihydroboron, BMS, Borane-dimethyl sulfide
About This Item
Recommended Products
form
liquid
Quality Level
reaction suitability
reagent type: reductant
density
0.801 g/mL at 25 °C (lit.)
functional group
thioether
storage temp.
2-8°C
SMILES string
B.CSC
InChI
1S/C2H6S.BH3/c1-3-2;/h1-2H3;1H3
InChI key
RMHDLBZYPISZOI-UHFFFAOYSA-N
Related Categories
General description
Application
- For the selective synthesis of 1,3,5-oxygenated compounds from dimethyl 3-oxoglutarate.
- For the conversion of ozonides to alcohols.
- In the CBS-catalyzed asymmetric reduction of ferrocenyl-1,3-diketones to 1,3-diols.
- For enantioselective reduction of ketones to chiral secondary alcohols in the presence of C3-symmetric tripodal hydroxyamide as a ligand.
- For the hydroboration reduction and other applications.
- With a dendrimeric supported L-pyrrolidinol in the asymmetric reduction of indanones and tetralones.
Packaging
Other Notes
Legal Information
signalword
Danger
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 3 Oral - Aquatic Chronic 3 - Eye Dam. 1 - Flam. Liq. 2 - Repr. 1B - Water-react 1
Storage Class
4.3 - Hazardous materials which set free flammable gases upon contact with water
wgk_germany
WGK 1
flash_point_f
64.4 °F
flash_point_c
18 °C
ppe
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![9-Borabicyclo[3.3.1]nonane solution 0.5 M in THF](/deepweb/assets/sigmaaldrich/product/structures/180/891/8b64e597-269d-4780-98b6-40889dfd06b9/640/8b64e597-269d-4780-98b6-40889dfd06b9.png)