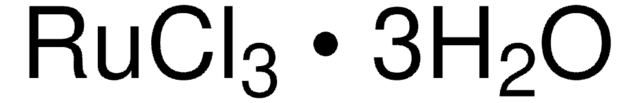206229
Ruthenium(III) chloride hydrate
ReagentPlus®
Synonym(s):
Ruthenium trichloride
About This Item
Recommended Products
Quality Level
product line
ReagentPlus®
form
powder and chunks
composition
Degree of hydration, ≤1
Ruthenium content, 40.00-49.00%
reaction suitability
reagent type: catalyst
core: ruthenium
impurities
≤0.1% Insoluble matter (C=1%, 25% HCl)
SMILES string
O.Cl[Ru](Cl)Cl
InChI
1S/3ClH.H2O.Ru/h3*1H;1H2;/q;;;;+3/p-3
InChI key
BIXNGBXQRRXPLM-UHFFFAOYSA-K
Looking for similar products? Visit Product Comparison Guide
General description
Application
Features and Benefits
Legal Information
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Chronic 2 - Eye Dam. 1 - Skin Corr. 1B
Storage Class
8A - Combustible corrosive hazardous materials
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Hydrogen is one of the most important resources in providing food, fuel, and chemical products for our everyday life. Sustainable catalytic hydrogen production from bioethanol has gained significant attention in recent years due to globally diminishing fossil fuel supplies, which have necessitated the search for new chemical feedstocks.
The prevailing strategies for heat and electric-power production that rely on fossil and fission fuels are having a negative impact on the environment and on our living conditions.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service