471232
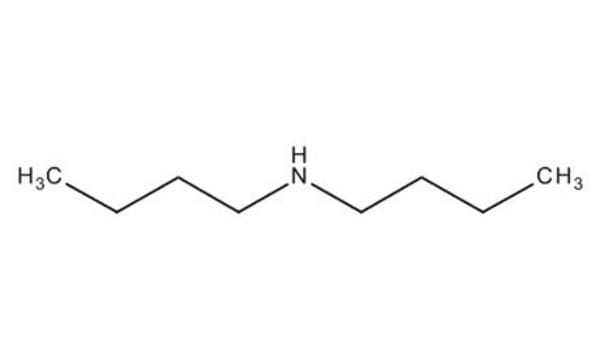
Dibutylamine
≥99.5%
Synonym(s):
N,N-Di-n-butylamine, N-Butyl-1-butanamine, n-Dibutylamine, Di-n-butylamine
About This Item
Recommended Products
vapor density
4.46 (vs air)
Quality Level
vapor pressure
1.9 mmHg ( 20 °C)
assay
≥99.5%
form
liquid
autoignition temp.
594 °F
expl. lim.
10 %
refractive index
n20/D 1.417 (lit.)
pH
11.1 (20 °C, 1 g/L)
bp
159 °C (lit.)
mp
−62 °C (lit.)
solubility
water: soluble 3.8 g/L at 20 °C
density
0.767 g/mL at 25 °C (lit.)
functional group
amine
SMILES string
CCCCNCCCC
InChI
1S/C8H19N/c1-3-5-7-9-8-6-4-2/h9H,3-8H2,1-2H3
InChI key
JQVDAXLFBXTEQA-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- N, N
- -Dibutyl-4-methylaniline by Pd-catalyzed Buchwald-Hartwig amination reaction with aryl chlorides and bromides.
- N,N-Dibutylacetamide by acetylation reaction with isopropenyl acetate.
It can also be used as:
- A strong hindered base in the different elimination reactions.
- An organocatalyst to synthesize annulated pyrano[2,3-d]pyrimidine derivatives via multicomponent condensation reaction of aromatic aldehydes, malononitrile, and barbituric acid.
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 2 Inhalation - Acute Tox. 3 Dermal - Acute Tox. 4 Oral - Eye Dam. 1 - Flam. Liq. 3 - Skin Corr. 1A
Storage Class
3 - Flammable liquids
wgk_germany
WGK 1
flash_point_f
104.9 °F - closed cup
flash_point_c
40.5 °C - closed cup
ppe
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service