All Photos(1)
About This Item
Empirical Formula (Hill Notation):
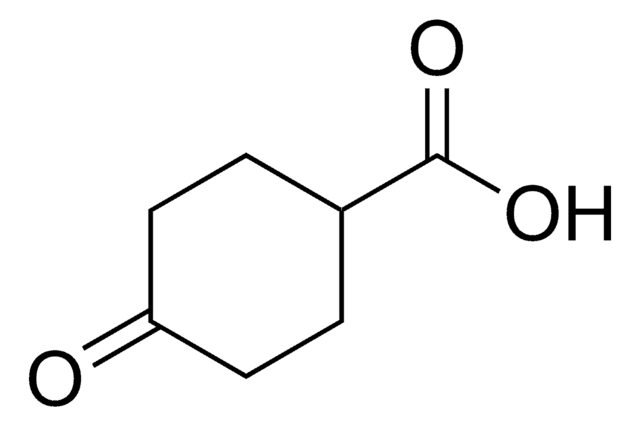
C6H8O3
CAS Number:
Molecular Weight:
128.13
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
97%
mp
59-62 °C (lit.)
functional group
carboxylic acid
ketone
SMILES string
OC(=O)C1CCC(=O)C1
InChI
1S/C6H8O3/c7-5-2-1-4(3-5)6(8)9/h4H,1-3H2,(H,8,9)
InChI key
RDSNBKRWKBMPOP-UHFFFAOYSA-N
Related Categories
General description
3-Oxo-1-cyclopentanecarboxylic acid , also known as 3-oxocyclopentanecarboxylic acid, is a keto acid derivative. It undergoes Curtius rearrangement with diphenyl phosphoryl azide and triethylamine in tert-butanol to form the corresponding boc-protected 1-(3-oxo)urea derivative.
Application
3-Oxo-1-cyclopentanecarboxylic acid may be used in the preparation of 3-hydroxycyclopentanecarboxylic acid via hydrogenation.
Substrate used in a study of biohydroxylation with mutants of cytochrome P450 BM-3.
Legal Information
Product of Rieke Metals, Inc.
Rieke is a registered trademark of Rieke Metals, Inc.
Rieke is a registered trademark of Rieke Metals, Inc.
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Studies of Configuration. V. The Preparation and Configuration of cis-3-Methoxycyclopentanecarboxylic Acid.
Noyce D and Fessenden J.
The Journal of Organic Chemistry, 24(5), 715-717 (1959)
Dieter F Münzer et al.
Chemical communications (Cambridge, England), (20), 2597-2599 (2005-05-19)
Substrate engineered, achiral carboxylic acid derivative was biohydroxylated with various mutants of cytochrome P450 BM-3 to give two out of the four possible diastereoisomers in high de and ee. The BM-3 mutants exhibit up to 9200 total turnovers for hydroxylation
Boc-protected 1-(3-oxocycloalkyl) ureas via a one-step Curtius rearrangement: mechanism and scope.
Sun X, et al.
Tetrahedron Letters, 55(4), 842-844 (2014)
Articles
Phase I biotransformation reactions increase drug compound polarity, mainly occurring in hepatic circulation.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service