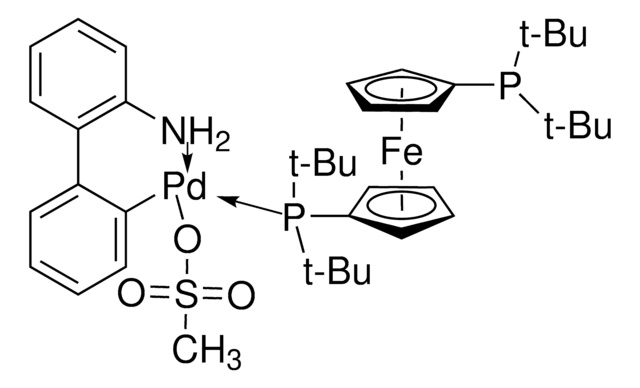
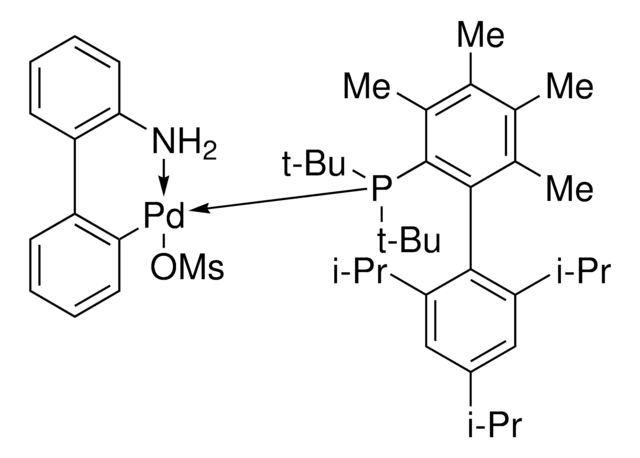
764183
APhos Pd G3
97%
Synonym(s):
APhos-Pd-G3, Palladium G3-(4-(N,N-Dimethylamino)phenyl)di-tert-butylphosphine, [4-(Di-tert-butylphosphino)-N,N-dimethylaniline-2-(2′-aminobiphenyl)]palladium(II) methanesulfonate
About This Item
Recommended Products
Quality Level
assay
97%
form
solid
feature
generation 3
reaction suitability
core: palladium
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: catalyst
reaction type: Cross Couplings
greener alternative product score
old score: 5
new score: 3
Find out more about DOZN™ Scoring
greener alternative product characteristics
Waste Prevention
Atom Economy
Less Hazardous Chemical Syntheses
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
mp
192-201 °C (decomposition)
functional group
phosphine
greener alternative category
SMILES string
NC1=C(C=CC=C1)C2=C([Pd]OS(C)(=O)=O)C=CC=C2.CN(C)C3=CC=C(C=C3)P(C(C)(C)C)C(C)(C)C
InChI
1S/C16H28NP.C12H10N.CH4O3S.Pd/c1-15(2,3)18(16(4,5)6)14-11-9-13(10-12-14)17(7)8;13-12-9-5-4-8-11(12)10-6-2-1-3-7-10;1-5(2,3)4;/h9-12H,1-8H3;1-6,8-9H,13H2;1H3,(H,2,3,4);/q;;;+1/p-1
InChI key
SNUBBUQVCDWEAV-UHFFFAOYSA-M
General description
Application
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
All of the preformed catalysts used in the kit are air and moisture stable complexes in their commercially available form.
Tools aid in kit setup for organic chemistry techniques, ensuring ease and success.
Materials Included in your KITALYSIS-24PD-2PK High-Throughput Screening Kit
G3 and G4 Buchwald palladium precatalysts are the newest air, moisture, and thermally stable crossing-coupling complexes used in bond formation for their versatility and high reactivity.
Related Content
The Buchwald group has developed a series of highly active and versatile palladium precatalysts and biarylphosphine ligands used in cross-coupling reactions for the formation of C-C, C–N, C–O, C–F, C–CF3, and C–S bonds. The ligands are electron-rich, and highly tunable to provide catalyst systems with a diverse scope, high stability and reactivity. Furthermore, the new series of precatalysts are air-, moisture and thermally-stable and display good solubility in common organic solvents. The use of precatalysts ensures the efficient generation of the active catalytic species and allows one to accurately adjust the ligand:palladium ratio. The ligands, precatalysts and methodology developed in the Buchwald group are user friendly and have rendered previously difficult cross couplings reactions, much easier to achieve.
Explore reliable, premium grade catalysis materials for your pharma or industrial project. Specialty chemicals and formulations are available in bulk quantities and volumes from a few grams to multi-metric tons with complete documentation to simplify your leap from development to commercialization.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service



![[1,1′-Bis(di-tert-butylphosphino)ferrocene]dichloropalladium(II) 98%](/deepweb/assets/sigmaaldrich/product/structures/192/459/02d1239c-1119-49d9-b392-a04d8f53855c/640/02d1239c-1119-49d9-b392-a04d8f53855c.png)