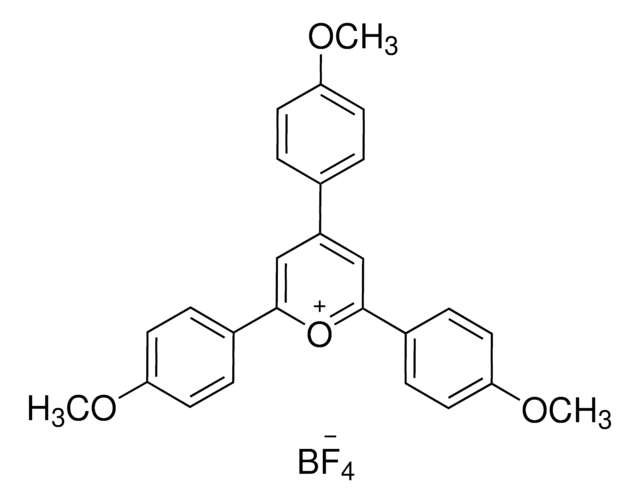
900685
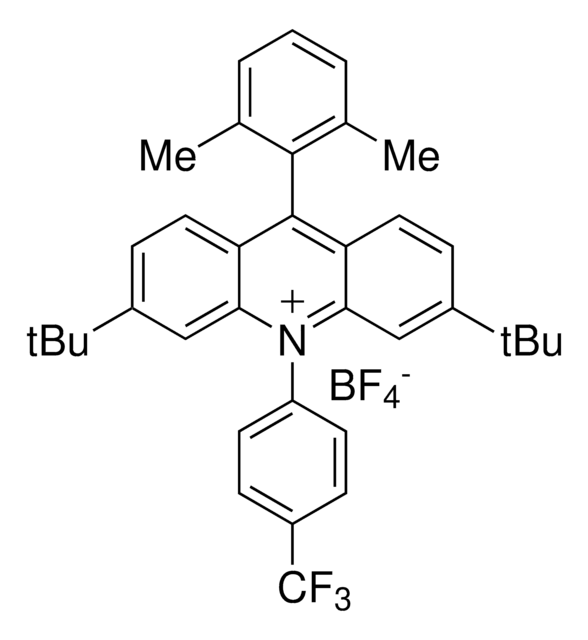
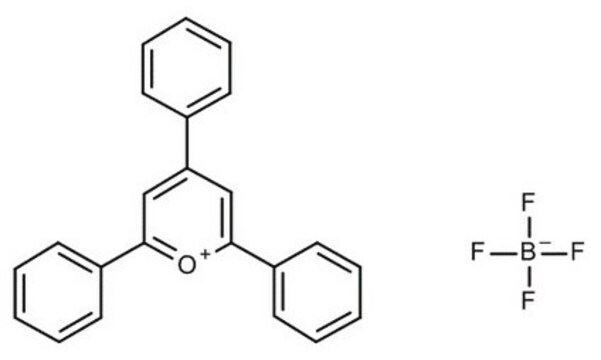
2,4,6-Tri(p-tolyl)pyrylium tetrafluoroborate salt
≥95%
Select a Size
Select a Size
About This Item
Recommended Products
Quality Level
assay
≥95%
form
powder
reaction suitability
reagent type: catalyst
reaction type: Photocatalysis
mp
298-303 °C
SMILES string
[B-](F)(F)(F)F.[o+]1c(cc(cc1c4ccc(cc4)C)c3ccc(cc3)C)c2ccc(cc2)C
InChI
1S/C26H23O.BF4/c1-18-4-10-21(11-5-18)24-16-25(22-12-6-19(2)7-13-22)27-26(17-24)23-14-8-20(3)9-15-23;2-1(3,4)5/h4-17H,1-3H3;/q+1;-1
InChI key
BWOXOWBZXLPNEN-UHFFFAOYSA-N
Related Categories
Application
Product can be used with our line of photoreactors: Including Penn PhD (Z744035) & SynLED 2.0 (Z744080)
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Related Content
Nicewicz lab focuses on reactive cation radical species for selective reactions, advancing synthesis methodologies for various functional groups.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service


![[Ir(dFCF3ppy)2-(5,5’-dCF3bpy)]PF6 ≥95%](/deepweb/assets/sigmaaldrich/product/structures/422/901/e00f3148-fb86-4f94-9e79-21d064c3f327/640/e00f3148-fb86-4f94-9e79-21d064c3f327.png)