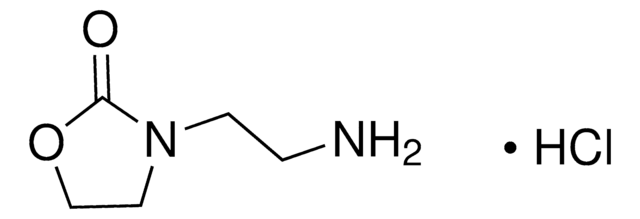
O9409
2-Oxazolidinone
98%
Synonym(s):
1,3-Oxazolidin-2-one, 2-Oxazolidone, 2-Oxo-1,3-oxazolidine, 2-Oxotetrahydro-1,3-oxazole
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Empirical Formula (Hill Notation):
C3H5NO2
CAS Number:
Molecular Weight:
87.08
Beilstein/REAXYS Number:
106251
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
98%
form
solid
bp
220 °C/48 mmHg (lit.)
mp
83-87 °C (lit.)
SMILES string
O=C1NCCO1
InChI
1S/C3H5NO2/c5-3-4-1-2-6-3/h1-2H2,(H,4,5)
InChI key
IZXIZTKNFFYFOF-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
2-Oxazolidinone can be employed as a precursor for the preparation of acryloyloxazolidin-2-ones , trans-3-cinnamoyloxazolidin-2-one, 3-bromo-2-oxazolidinone , 4-methoxy-2-oxazolidinone , coordination compound, tetrakis[μ-(2-oxazolidinonato-κN3:κO2)]tetra-, (4Cu-Cu).
Storage Class
13 - Non Combustible Solids
wgk_germany
WGK 3
ppe
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Cysteine-Aminoethylation-Assisted Chemical Ubiquitination of Recombinant Histones
Chu G-C, et al.
Journal of the American Chemical Society, 141(8), 3654-3663 (2019)
Nada A Al-Hasawi et al.
Anticancer research, 40(9), 5125-5140 (2020-09-04)
Neuroblastoma (NB), the most common extracranial malignant childhood tumor accounts for about 15% of cancer-related deaths in children. Despite the intensive treatment of patients with high-risk scarification of NB, clinical outcomes indicate tumor recurrence greater than 50% and late severe
Mathieu Esgulian et al.
Organic & biomolecular chemistry, 15(6), 1453-1462 (2017-01-21)
A number of cyclic derivatives of 3-amino-2,4-dihydroxybutanoic acid are known in the literature but they are often prepared from other cyclic precursors. This study showed that the title compound too may serve as a convenient substrate for cyclization reactions. Using
Stereocontrolled Synthesis of trans/cis-2, 3-Disubstituted Cyclopropane-1, 1-diesters and Applications in the Syntheses of Furanolignans
Shen Y, et al.
The Journal of Organic Chemistry, 83(20), 12549-12558 (2018)
Photoinduced, copper-catalyzed alkylation of amides with unactivated secondary alkyl halides at room temperature
Do Hien-Quang, et al.
Journal of the American Chemical Society, 136(5), 2162-2167 (2014)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service