8.22288
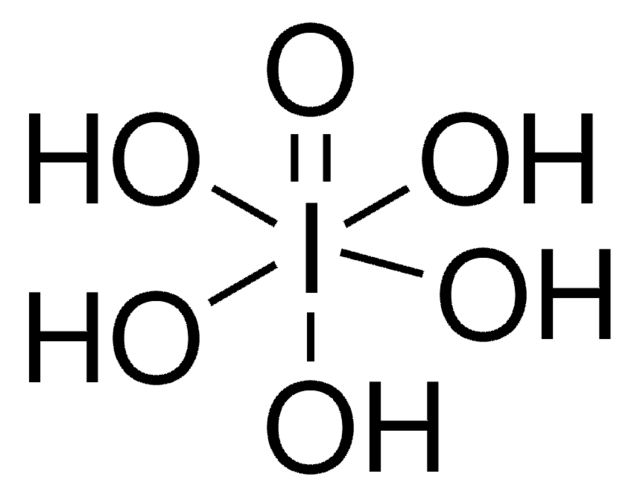
Periodic acid
for synthesis
Synonym(s):
Periodic acid
About This Item
Recommended Products
Quality Level
assay
≥98% (iodometric)
form
solid
reaction suitability
reagent type: oxidant
pH
1.2 (20 °C, 100 g/L in H2O)
mp
122 °C
solubility
ethanol: soluble at 20 °C
water: soluble at 20 °C
bulk density
1400 kg/m3
storage temp.
15-25°C
InChI
1S/H5IO6/c2-1(3,4,5,6)7/h(H5,2,3,4,5,6,7)
InChI key
TWLXDPFBEPBAQB-UHFFFAOYSA-N
Application
It can also be used:
- In the selective N-nitrosation of secondary amines in the presence of sodium nitrite, and wet silica gel.
- As a stoichiometric oxidant with chromium (IV) acetate for the oxidation of tertiary C−H bonds to tertiary alcohols.
- In combination with chromium oxide to oxidize primary alcohols to carboxylic acids; alkylbenzenes to benzoic acids; and cyclic benzyl ethers to the corresponding lactones.
- In the iodination of bulky polyalkyl benzenes in the presence of iodine.
- In the oxidative coupling of polycyclic aromatic hydrocarbons in the presence of aqueous acetic acid.
- In the selective oxidation of sulfides to sulfoxides using FeCl3 as a catalyst.
Analysis Note
Melting range (lower value): ≥ 124 °C
Melting range (upper value): ≤ 129 °C
signalword
Danger
Hazard Classifications
Aquatic Acute 1 - Aquatic Chronic 1 - Eye Dam. 1 - Ox. Sol. 1 - Skin Corr. 1B - STOT RE 1 Oral
target_organs
Thyroid
Storage Class
5.1A - Strongly oxidizing hazardous materials
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service