General description
AMP-activated protein kinase alpha subunit, isoform A (EC 2.7.11.16; UniProt O18645; also known as AMP-activated protein kinase alpha subunit, isoform B, AMP-activated protein kinase alpha subunit, isoform C, EG:132E8.2 protein, FI03728p, SNF1A/AMP-activated protein kinase) is encoded by the AMPKalpha (also known as EG:132E8.2, SNF1A, SNF1A-RA) gene (ORF CG3051/Dmel_CG3051; Gene ID 43904) in Drosophila melanogaster (Fruit fly) species. The yeast sucrose non-fermenting 1 (SNF1) and the AMP-activated protein kinase (AMPK) in drosphila and mammals are orthologous heterotrimeric serine-threonine kinases, each consisting of a catalytic α subunit and regulatory β and γ subunits, that sense decreased cellular energy levels when the AMP to ATP ratio rises due to a high rate of metabolism or decreased nutrient availability. AMP binding to the γ subunit results in an enhanced phosphorylation of the catalytic α subunit on an activation loop threonine and AMPK allosteric activation. Activated AMPK phosphorylates downstream targets, leading to energy conservation and inhibition of ATP consumption. For example, the high rate of ATP use in contracting muscle causes AMPK-mediated phosphorylation/inactivation of acetyl-CoA carboxylase (ACC), thereby increasing fatty acid oxidation and restoring cellular energy. AMPK functions as a positive regulator of autophagy by phosphorylating the protein kinase ULK1 that initiates autophagy as well as by inhibiting the mammalian TOR (mTOR) pathway activity via phosphorylating mTOR-binding partner raptor. In mammals, there exist two different α (encoded by the PRKAA1 and PRKAA2 gene), two different β (encoded by the PRKAB1 and PRKAB2 gene), and three different γ (encoded by the PRKAG1, PRKAG2, and PRKAG3 gene) subunits.
Specificity
Clone 34.2 reacts with drosophila AMPK alpha subunit, as well as AMPK catalytic subunits alpha 1 and alpha 2 of diverse mammalian species, including human, rat, and mouse.
Immunogen
Epitope: N-terminal catalytic domain.
Linear peptide corresponding to a sequence from the N-terminal catalytic domain of drosophila AMPK alpha subunit.
Application
Anti-AMPK alpha Antibody, clone 34.2 is an antibody against AMPK alpha 1/2 for use in Western Blotting.
Research Category
Signaling
Research Sub Category
Kinases & Phosphatases
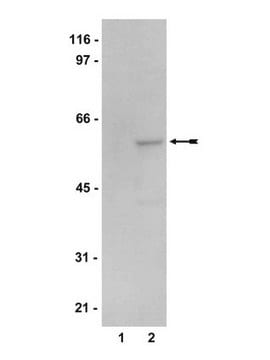
Western Blotting Analysis: A representative lot detected AMPK alpha 1/2 in HEK293 cell lysate, rat brain homogenate, and Drosophila melanogaster S2 cell lysate (Courtesy of Professor Jay Brenman, UNC School of Medicine, USA).
Quality
Evaluated by Western Blotting in HepG2 cell lysate.
Western Blotting Analysis: 1.0 µg/mL of this antibody detected AMPK alpha 1/2 in 10 µg of HepG2 cell lysate.
Target description
~73 kDa observed. 64.60 kDa (drosophila), 64.01/63.93/63.97 kDa (human/mouse/rat AMPK subunit alpha-1), 62.32/62.02/62.26 kDa (human/mouse/rat AMPK subunit alpha-2) calculated.
Physical form
Format: Purified
Protein G purified.
Purified mouse monoclonal IgG2aκ antibody in buffer containing 0.1 M Tris-Glycine (pH 7.4), 150 mM NaCl with 0.05% sodium azide.
Storage and Stability
Stable for 1 year at 2-8°C from date of receipt.
Other Notes
Concentration: Please refer to lot specific datasheet.
Disclaimer
Unless otherwise stated in our catalog or other company documentation accompanying the product(s), our products are intended for research use only and are not to be used for any other purpose, which includes but is not limited to, unauthorized commercial uses, in vitro diagnostic uses, ex vivo or in vivo therapeutic uses or any type of consumption or application to humans or animals.