49603
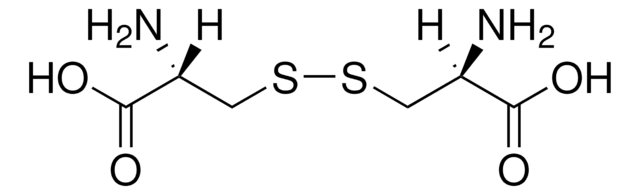
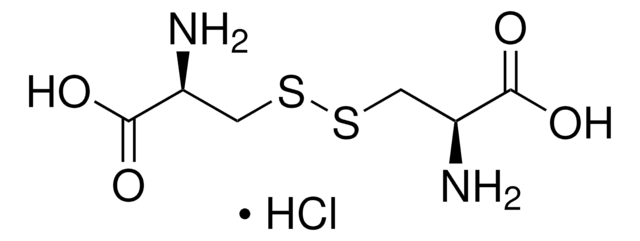
L-Cystine
certified reference material, TraceCERT®, Manufactured by: Sigma-Aldrich Production GmbH, Switzerland
Synonym(s):
(R,R)-3,3′-Dithiobis(2-aminopropionicacid)
About This Item
Recommended Products
grade
certified reference material
TraceCERT®
Quality Level
product line
TraceCERT®
analyte chemical class(es)
amino acids, peptides, proteins
manufacturer/tradename
Manufactured by: Sigma-Aldrich Production GmbH, Switzerland
storage condition
under inert gas
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
mp
>240 °C (dec.) (lit.)
application(s)
cleaning products
cosmetics
flavors and fragrances
food and beverages
personal care
format
neat
SMILES string
N[C@@H](CSSC[C@H](N)C(O)=O)C(O)=O
InChI
1S/C6H12N2O4S2/c7-3(5(9)10)1-13-14-2-4(8)6(11)12/h3-4H,1-2,7-8H2,(H,9,10)(H,11,12)/t3-,4-/m0/s1
InChI key
LEVWYRKDKASIDU-IMJSIDKUSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Certified content by quantitative NMR incl. uncertainty and expiry date are given on the certificate.
Download your certificate at: http://www.sigma-aldrich.com.
Check out the entire portfolio of our amino acids certified reference materials (CRM)
Application
Packaging
Legal Information
Storage Class
11 - Combustible Solids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Certificates of Analysis (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documents section.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service