82800
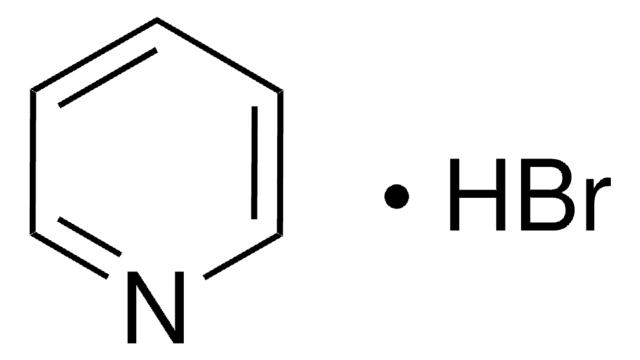
Pyridine hydrochloride
purum, ≥98.0% (AT)
Synonym(s):
Pyridinium chloride
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C5H5N · HCl
CAS Number:
Molecular Weight:
115.56
Beilstein/REAXYS Number:
3615340
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
grade
purum
Quality Level
assay
≥98.0% (AT)
form
powder
bp
222-224 °C (lit.)
mp
143-147 °C
145-147 °C (lit.)
SMILES string
Cl[H].c1ccncc1
InChI
1S/C5H5N.ClH/c1-2-4-6-5-3-1;/h1-5H;1H
InChI key
AOJFQRQNPXYVLM-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Pyridine hydrochloride can be used:
- As an acid-base catalyst in the conversion of 4-pyridyl propargylic alcohols to the (E)-propenones and propynones.
- As a reagent in the synthesis of 2-arylindene-1-ones , baicalein , pinosylvin derivatives.
- As a reagent in O-demethylation reaction under microwave irradiation.
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Synthesis and inhibitory effects of pinosylvin derivatives on prostaglandin E2 production in lipopolysaccharide-induced mouse macrophage cells
Park E-J, et al.
Bioorganic & medicinal chemistry letters, 14(23), 5895-5898 (2004)
Recent advances in ether dealkylation
Weissman SA and Zewge D
Tetrahedron, 61(33), 7833-7863 (2005)
Total synthesis of baicalein
Chen D-Z, et al.
Journal of Asian natural products research, 12(2), 124-128 (2010)
Estrogen receptor ligands: design and synthesis of new 2-arylindene-1-ones
McDevitt RE, et al.
Bioorganic & Medicinal Chemistry Letters, 15(12), 3137-3142 (2005)
Facile conversion of pyridine propargylic alcohols to enones: stereochemistry of protonation of allenol
Erenler R, et al.
Tetrahedron Letters, 46(34), 5683-5685 (2005)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service