C1131
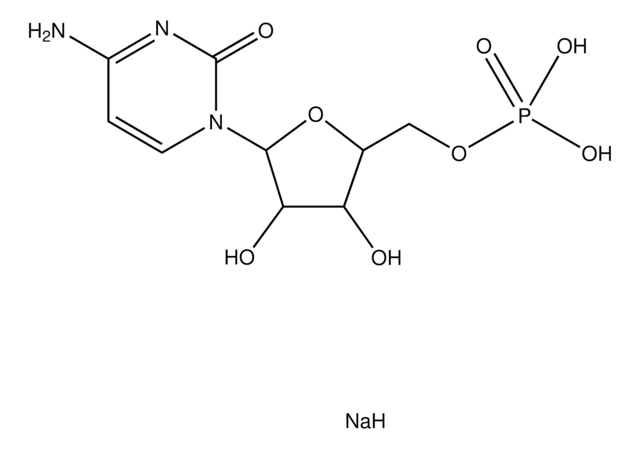
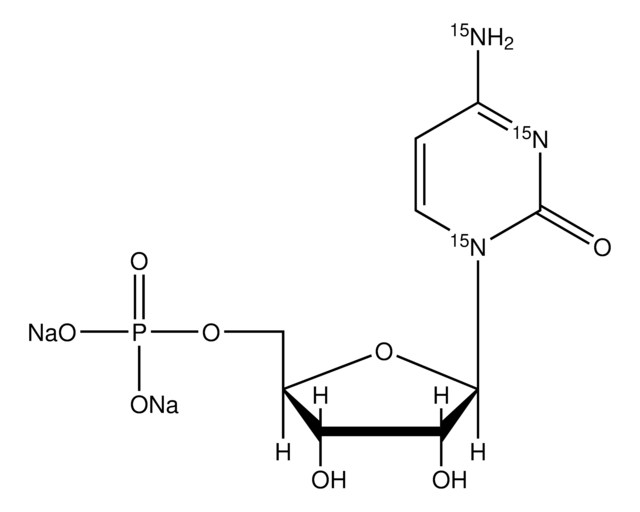
Cytidine 5′-monophosphate
Sigma Grade, ≥99% (HPLC), synthetic, powder
Synonym(s):
5′-CMP, 5′-Cytidylic acid, C-5′-P
About This Item
Recommended Products
biological source
synthetic
Quality Level
grade
Sigma Grade
assay
≥99% (HPLC)
form
powder
solubility
1 M NH4OH: 50 mg/mL, clear, colorless
storage temp.
2-8°C
SMILES string
NC1=NC(=O)N(C=C1)[C@@H]2O[C@H](COP(O)(O)=O)[C@@H](O)[C@H]2O
InChI
1S/C9H14N3O8P/c10-5-1-2-12(9(15)11-5)8-7(14)6(13)4(20-8)3-19-21(16,17)18/h1-2,4,6-8,13-14H,3H2,(H2,10,11,15)(H2,16,17,18)/t4-,6-,7-,8-/m1/s1
InChI key
IERHLVCPSMICTF-XVFCMESISA-N
Looking for similar products? Visit Product Comparison Guide
Application
Biochem/physiol Actions
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
ZIC®-cHILIC is a densely bonded zwitterionic stationary phase with phosphorylcholine functional groups covalently attached to silica.
HILIC separation is an alternative that permits sensitive MS detection and without the use of ion-pair reagents.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service