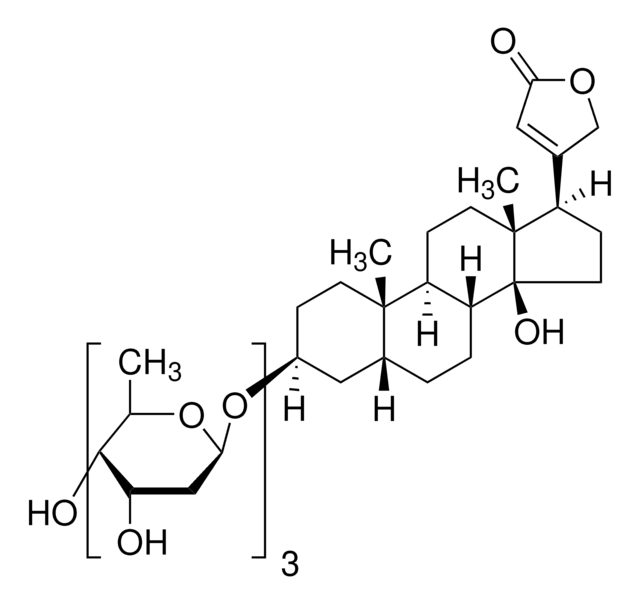
D141
Digitonin
Used as non-ionic detergent
Synonym(s):
Digitin
About This Item
Recommended Products
biological source
plant seeds (Digitalis purpurea)
Quality Level
form
powder
optical activity
[α]20/D −54°, c = 2.8 in methanol(lit.)
mol wt
micellar avg mol wt 70,000
aggregation number
60
impurities
<6.0% water
CMC
<0.5 mM (20-25°C)
<0.5 mM (20-25°C)
mp
230-240 °C (dec.) (lit.)
solubility
H2O: ~5 % (w/v) (solubilized by heating to 95 °C - 98 °C and then cooling to room temp.)
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Moreover, Digitonin selectively permeabilizes the cholesterol-rich plasma membrane, leaving organelle membranes intact. This feature facilitates the extraction and purification of specific organelles, contributing to the study of their functions and roles in cellular processes. The detergent′s capability to permeabilize diverse cell types allows the study of intracellular components and processes by introducing molecules, antibodies, or enzymes into cells. Additionally, Digitonin finds application in immunocytochemistry experiments, enabling the labeling and detection of intracellular proteins and structures.
Application
Biochem/physiol Actions
Features and Benefits
Quality
Other Notes
comparable product
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 3 Oral - STOT RE 2
Storage Class
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
This page shows how to solubilize membrane proteins with products from Cytiva.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service